Biopolymers vs Petroleum Plastics: A Comprehensive Life Cycle Assessment for Sustainable Biomedical Research
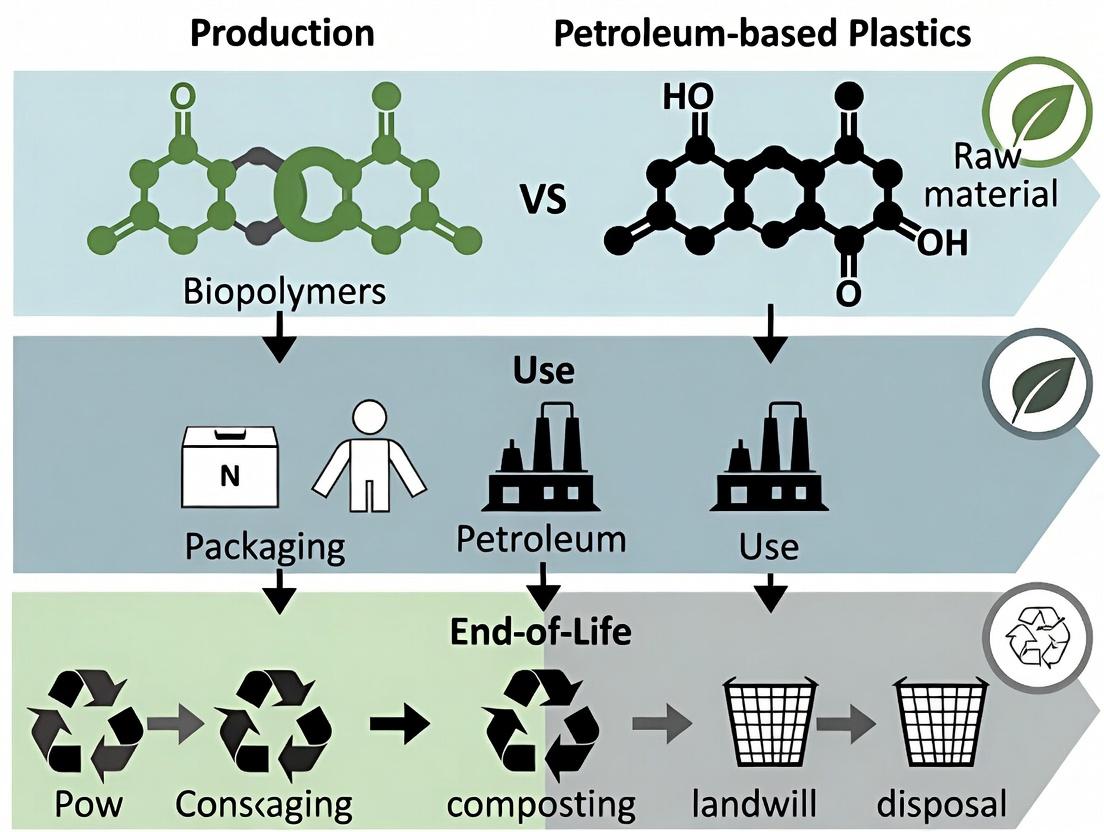
This article provides a detailed Life Cycle Assessment (LCA) framework comparing biopolymers and conventional petroleum-based plastics, specifically tailored for researchers and drug development professionals.
Biopolymers vs Petroleum Plastics: A Comprehensive Life Cycle Assessment for Sustainable Biomedical Research
Abstract
This article provides a detailed Life Cycle Assessment (LCA) framework comparing biopolymers and conventional petroleum-based plastics, specifically tailored for researchers and drug development professionals. We explore the fundamental definitions and sourcing of these materials, establish robust LCA methodologies for biomedical applications, address critical challenges in sterilization and material stability, and present a validated comparative analysis of their environmental impacts, biocompatibility, and performance in clinical settings. The synthesis aims to inform sustainable material selection in pharmaceutical packaging, medical devices, and therapeutic delivery systems.
Defining the Contenders: Sourcing, Chemistry, and Key Biopolymers for Biomedical Use
What are Biopolymers? From PLA and PHA to Starch and Chitosan
Biopolymers are polymers produced by living organisms or derived from renewable biomass sources. Within the context of life cycle assessment (LCA) research comparing biopolymers to petroleum-based plastics, they present a complex sustainability profile. This guide objectively compares the performance of prominent biopolymers—Polylactic Acid (PLA), Polyhydroxyalkanoates (PHA), Starch-based polymers, and Chitosan—against conventional plastics like Polyethylene (PE) and Polyethylene Terephthalate (PET). Performance is evaluated through key properties critical for packaging, biomedical, and pharmaceutical applications.
Mechanical & Thermal Property Comparison
Table 1: Comparative material properties of biopolymers and conventional plastics. Data synthesized from recent tensile, DSC, and TGA studies.
| Polymer | Tensile Strength (MPa) | Elongation at Break (%) | Glass Transition Temp. Tg (°C) | Degradation Temp. (Td, °C) | Key Limitation |
|---|---|---|---|---|---|
| PLA | 50-70 | 2-10 | 55-60 | 300-350 | Brittle, low toughness |
| PHA (PHB) | 20-40 | 3-8 | 0-5 | 250-300 | Thermally unstable, brittle |
| Starch-based | 5-30 | 20-100 | 50-60 (dry) | 220-300 | Highly hydrophilic |
| Chitosan | 30-60 | 5-30 | ~120 | 220-300 | Soluble in acidic solutions |
| PET | 55-75 | 50-300 | 70-80 | ~400 | High energy to produce |
| LDPE | 8-20 | 100-900 | -120 | ~400 | Fossil-derived |
Environmental Degradation Performance
Table 2: Biodegradation data under controlled composting (ISO 14855) and marine conditions (ASTM D6691).
| Polymer | Time for >90% Mineralization (Industrial Compost) | Marine Degradation (6 months) | Key Degradation Mechanism |
|---|---|---|---|
| PLA | 90-180 days | <5% mass loss | Hydrolysis then microbial assimilation |
| PHA | 40-100 days | 30-80% mass loss | Direct enzymatic degradation |
| Starch-based | 30-60 days | 10-50% mass loss | Swelling, microbial enzymatic attack |
| Chitosan | 50-100 days | 20-60% mass loss | Lysozyme-mediated hydrolysis |
| PET | No significant degradation | No significant degradation | Photodegradation only over decades |
| LDPE | No significant degradation | <2% mass loss | Fragmentative, not assimilative |
Experimental Protocol: Standard Biodegradation Test (ISO 14855)
- Sample Preparation: Precisely weigh 10g of test polymer film (thickness: 100±20 µm) and grind to particles <2mm.
- Inoculum: Prepare mature compost from biowaste, sieved to <10mm, with a volatile solids content >30%.
- Reactor Setup: Mix test material with 600g of wet compost in a 2L bioreactor. Maintain at 58°C ±2°C in a controlled incubator.
- Control & Blank: Set up positive control (cellulose) and blank (compost only) reactors in triplicate.
- Aeration & Monitoring: Continuously aerate with CO₂-free, humidified air. Measure evolved CO₂ in the effluent gas via NaOH trap titration or IR analysis weekly.
- Calculation: Calculate the percentage biodegradation as: (CO₂ from test - CO₂ from blank) / (Theoretical CO₂ of test material) x 100.
Barrier & Functional Properties
Table 3: Barrier properties critical for packaging and drug delivery.
| Polymer | Oxygen Permeability (cm³·mm/m²·day·atm) | Water Vapor Permeability (g·mm/m²·day·atm) | Key Functional Attribute |
|---|---|---|---|
| PLA | 15-25 | 15-25 | High clarity, printable |
| PHA | 5-15 | 10-20 | Good UV resistance, biocompatible |
| Starch-based | 500-1000 | 200-500 | High O₂ scavenging potential |
| Chitosan | 0.5-5 | 500-1000 | Antimicrobial, film-forming |
| PET | 2-5 | 1-3 | Excellent barrier, rigid |
| LDPE | 400-800 | 1-2 | Excellent moisture barrier |
Diagram 1: LCA research workflow.
Diagram 2: Biodegradation vs. fragmentation pathways.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential materials and reagents for biopolymer research and characterization.
| Reagent/Material | Function in Research | Example Use Case |
|---|---|---|
| Chloroform | Solvent for extracting and processing PHA from bacterial biomass. | PHA purification for film casting. |
| Lipase (from Pseudomonas sp.) | Enzyme for catalyzing hydrolysis of polyesters (PHA, PLA). | Standardized enzymatic degradation assay. |
| Lysozyme | Enzyme that catalyzes hydrolysis of chitosan glycosidic bonds. | Studying chitosan film biodegradation in biomedical contexts. |
| α-Amylase & Glucoamylase | Enzymes for starch hydrolysis. | Quantifying biodegradability of starch-based blends. |
| Simulated Body Fluid (SBF) | Ionic solution mimicking blood plasma. | Testing bioactivity and degradation of polymers for drug delivery. |
| Thermogravimetric Analyzer (TGA) | Instrument measuring mass loss as function of temperature. | Determining thermal stability and degradation temperature (Td). |
| Differential Scanning Calorimeter (DSC) | Instrument measuring heat flows associated with phase transitions. | Measuring Glass Transition (Tg) and Melting (Tm) temperatures. |
| Tensile Testing Machine | Instrument applying uniaxial tension to sample. | Determining tensile strength and elongation at break (ASTM D638). |
Within the framework of life cycle assessment (LCA) research comparing biopolymers to petroleum-based plastics, understanding the foundational production and performance of conventional polymers is critical. This guide objectively compares the performance of polypropylene (PP), polyethylene terephthalate (PET), and polyvinyl chloride (PVC) against common biopolymer alternatives, supported by experimental data.
Performance Comparison: Mechanical and Thermal Properties
The following table summarizes key performance metrics from recent experimental studies comparing conventional plastics with polylactic acid (PLA) and polyhydroxyalkanoates (PHA) as benchmark biopolymers.
Table 1: Mechanical and Thermal Properties Comparison
| Property | PP | PET | PVC | PLA (Biopolymer) | PHA (Biopolymer) | Test Standard |
|---|---|---|---|---|---|---|
| Tensile Strength (MPa) | 25-40 | 55-75 | 40-60 | 50-70 | 25-40 | ASTM D638 |
| Young's Modulus (GPa) | 1.5-2.0 | 2.0-4.1 | 2.4-4.1 | 3.0-3.5 | 0.5-1.5 | ASTM D638 |
| Elongation at Break (%) | 100-600 | 50-150 | 20-40 | 4-10 | 5-15 | ASTM D638 |
| Glass Transition Temp. Tg (°C) | -10 to -20 | 70-80 | 70-85 | 55-60 | -30 to 10 | ASTM E1356 |
| Melting Temp. Tm (°C) | 160-175 | 245-265 | 100-260 (Decomp.) | 150-160 | 140-175 | ASTM D3418 |
Performance Comparison: Barrier and End-of-Life Properties
Barrier properties are crucial for packaging, while degradation profiles are central to LCA studies on environmental impact.
Table 2: Barrier Properties and Degradation Characteristics
| Property | PP | PET | PVC | PLA | PHA | Test Method |
|---|---|---|---|---|---|---|
| O2 Permeability (cm³·mm/m²·day·atm) | 100-200 | 3-6 | 5-20 | 15-20 | 10-15 | ASTM D3985 |
| Water Vapor Trans. Rate (g·mm/m²·day) | 0.4-0.6 | 1.0-1.5 | 2.0-5.0 | 15-20 | 10-20 | ASTM E96 |
| Hydrolysis Rate (Mass loss % / 30 days) | <1 | <1 | <1 | 40-60 (pH 10, 60°C) | 50-80 (Marine) | ASTM D6691 |
| Compost Degradation (Days to >90% min.) | >1000 | >1000 | >1000 | 60-90 (Industrial) | 40-70 (Industrial) | ASTM D6400 |
Experimental Protocols for Cited Data
Protocol 1: Tensile Strength and Modulus (ASTM D638)
- Specimen Preparation: Injection mold or cut dumbbell-shaped specimens (Type I) with defined gauge length.
- Conditioning: Condition specimens at 23 ± 2°C and 50 ± 5% relative humidity for 40+ hours.
- Testing: Mount specimen in universal testing machine. Apply tension at a constant crosshead speed of 5 mm/min for rigid plastics (e.g., PET, PLA) or 50 mm/min for ductile plastics (e.g., PP).
- Data Collection: Record force vs. elongation. Calculate tensile strength (max force/original area), elongation at break, and Young's Modulus (slope of linear elastic region).
Protocol 2: Compost Degradation (ASTM D6400)
- Material Preparation: Prepare test specimens (typically 25mm x 25mm x 1mm). Weigh initial mass (M0).
- Compost Environment: Place specimens in controlled composting vessels with mature, bioactive compost maintained at 58 ± 2°C and ~55% moisture.
- Aeration & Monitoring: Aerate with humidified air. Monitor CO2 evolution and compost chemistry.
- Recovery & Analysis: Recover specimens after predetermined intervals (e.g., 15, 30, 60 days). Clean, dry, and weigh final mass (Mf). Calculate percentage disintegration: ((M0 - Mf) / M0) * 100.
Pathway and Workflow Diagrams
Title: Petrochemical to Plastic Production Pathway
Title: LCA Experimental Workflow: Petro vs Bio Plastics
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Research Materials for Polymer LCA Studies
| Item | Function in Research |
|---|---|
| Universal Testing Machine (UTM) | Measures tensile, flexural, and compressive properties of plastic specimens under controlled forces. |
| Differential Scanning Calorimeter (DSC) | Determines thermal transitions (Tg, Tm, crystallization temperature) and percent crystallinity of polymer samples. |
| Gas Chromatograph-Mass Spectrometer (GC-MS) | Analyzes volatile organic compounds (VOCs) emitted during polymer processing or degradation. |
| Simulated Compost/Marine Media | Standardized biotic environments (per ASTM D6691/D6400) for assessing biodegradation rates under controlled lab conditions. |
| FT-IR Spectrometer | Identifies chemical functional groups, monitors degradation-induced chemical changes, and verifies polymer composition. |
| Gel Permeation Chromatograph (GPC) | Determines the molecular weight distribution and average molecular weights of polymers, critical for property correlation. |
| Accelerated Weathering Chamber | Simulates long-term environmental aging (UV, moisture, heat) to study polymer durability and degradation pathways. |
This comparison guide objectively evaluates the sourcing of raw materials for polymer production, a critical initial phase in the life cycle assessment of biopolymers versus petroleum-based plastics. The analysis focuses on environmental, energetic, and resource metrics relevant to researchers and industrial scientists.
Quantitative Comparison of Sourcing Metrics
Table 1: Comparative Data for Feedstock Sourcing (Per Functional Unit of Polymer Precursor)
| Metric | Fossil Fuel Extraction (e.g., Crude Oil for Naphtha) | Renewable Feedstock (e.g., Corn Starch for Glucose) | Data Source & Notes |
|---|---|---|---|
| Land Use (m²·yr/kg) | 0.05 - 0.2 (for drilling/refining) | 0.8 - 2.5 (for crop cultivation) | [1,2] Renewable feedstock requires agricultural land. |
| Water Consumption (L/kg) | 5 - 15 | 200 - 600 (irrigation) | [1,3] Highly dependent on crop type and region. |
| Non-Renewable Energy Use (MJ/kg) | 35 - 50 (inherent to feedstock) | 10 - 25 (for farming & processing) | [1,4] Fossil fuel embodies energy; renewable uses external inputs. |
| Global Warming Potential (kg CO₂-eq/kg) | 1.5 - 3.0 (from extraction/refining) | 0.5 - 1.5 (excluding carbon uptake) | [1,5] Assumes standard agricultural practices. |
| Feedstock Yield (ton/ha·yr) | N/A (finite reserve) | 4 - 8 (dry mass of starch) | [2] Fossil yield is a reserve depletion rate. |
| Elemental Purity (C, H, O) | High, but requires cracking | Lower, contains heteroatoms | Laboratory analytical data. |
Experimental Protocols for Feedstock Analysis
Protocol 1: Life Cycle Inventory (LCI) Data Collection for Feedstock Sourcing
- Goal & Scope Definition: Define the functional unit (e.g., 1 kg of purified polymer precursor). Set system boundaries from well-to-gate or farm-to-gate.
- Data Acquisition: For fossil feedstocks, collect primary data from refinery operations or secondary databases (e.g., Ecoinvent) on energy inputs, direct emissions, and land transformation. For renewable feedstocks, collect agricultural data on seeding, irrigation, fertilization, harvesting, and initial processing (e.g., milling corn).
- Allocation: For renewable systems, apply allocation (mass, economic, or energy-based) to partition environmental burdens between the primary product (e.g., grain) and co-products (e.g., stalk, husk).
- Impact Calculation: Use LCA software (e.g., SimaPro, GaBi) to calculate midpoint impact categories (Global Warming, Water Use, Land Use) for the defined functional unit.
Protocol 2: Proximate and Ultimate Analysis of Feedstocks
- Sample Preparation: Grind fossil (e.g., petroleum coke) and renewable (e.g., lignocellulosic biomass) samples to a uniform particle size (< 0.5 mm). Dry in an oven at 105°C for 24 hours.
- Proximate Analysis (ASTM D5142): Use a thermogravimetric analyzer (TGA). Record mass loss under inert atmosphere (N₂) to 900°C to determine volatile matter and fixed carbon. Subsequently, switch to oxidative atmosphere (air) to determine ash content.
- Ultimate Analysis (CHNS/O): Use an elemental analyzer (e.g., CHNS/O analyzer). For C, H, N, S, combust sample at high temperature with oxygen. Detect gases via thermal conductivity. Oxygen content is often calculated by difference.
- Data Interpretation: Compare C/H/O ratios and ash content to predict processing requirements and potential catalyst poisoning in subsequent chemical conversion steps.
Visualizations
Title: Feedstock Sourcing Pathways to Polymer Monomers
Title: LCA System Boundaries for Feedstock Sourcing
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Feedstock Sourcing Research
| Item / Reagent | Function in Research |
|---|---|
| Thermogravimetric Analyzer (TGA) | Determines thermal stability, moisture, volatile content, and ash composition of fossil and biomass feedstocks. |
| CHNS/O Elemental Analyzer | Quantifies carbon, hydrogen, nitrogen, sulfur, and oxygen content, critical for mass balance and stoichiometric calculations. |
| Life Cycle Inventory (LCI) Database (e.g., Ecoinvent, GREET) | Provides secondary data on background processes (e.g., fertilizer production, diesel combustion, grid electricity) for LCA modeling. |
| Enzymatic Hydrolysis Kits (Cellulase, Amylase) | Used in lab-scale saccharification experiments to determine the maximum reducing sugar yield from lignocellulosic or starchy biomass. |
| ICP-MS/OES Reagents & Standards | For preparing samples and calibrations to measure trace metal and catalyst poison concentrations (e.g., Na, K, Ca, heavy metals) in feedstocks. |
| Solvents for Soxhlet Extraction (e.g., Ethanol, Hexane) | Used to extract lipids, resins, and other non-structural compounds from biomass to assess purity and compositional variability. |
The selection of materials for biomedical applications—such as drug delivery systems, implantable devices, and diagnostic packaging—requires a stringent evaluation of key performance properties. Within the context of life cycle assessment (LCA) research comparing biopolymers to petroleum-based plastics, understanding the trade-offs in mechanical, optical, and protective characteristics is crucial for informed, sustainable material selection. This guide objectively compares common classes of materials based on recent experimental data.
Comparison of Key Properties for Biomedical Polymers
The following table summarizes quantitative data from recent studies (2022-2024) on critical properties. Data are presented as ranges to account for variations in polymer grades, processing, and testing methodologies.
Table 1: Comparative Material Properties for Biomedical Applications
| Material Class | Specific Polymer | Tensile Strength (MPa) | Young's Modulus (GPa) | Water Vapor Transmission Rate (WVTR) (g·mil/m²·day) | Oxygen Transmission Rate (OTR) (cm³·mil/m²·day·atm) | Haze / Clarity (% Haze) |
|---|---|---|---|---|---|---|
| Petro-based (Conventional) | Polypropylene (PP, medical grade) | 25 - 38 | 1.5 - 2.2 | 0.2 - 0.4 | 100 - 200 | 5 - 15 (Opaque to translucent) |
| Polyethylene Terephthalate (PET, oriented) | 55 - 75 | 2.7 - 4.1 | 1.0 - 1.5 | 3 - 10 | 1 - 4 (Clear) | |
| Polyvinyl Chloride (PVC, plasticized) | 10 - 25 | 0.01 - 0.5 | 2 - 8 | 50 - 200 | 2 - 10 (Clear) | |
| Bio-based / Biodegradable | Polylactic Acid (PLA) | 45 - 70 | 3.0 - 4.5 | 15 - 25 | 150 - 300 | 3 - 10 (Clear) |
| Polyhydroxyalkanoates (PHA, PHB type) | 25 - 40 | 3.5 - 4.0 | 10 - 20 | 20 - 50 | 20 - 80 (Hazy) | |
| Thermoplastic Starch (TPS) | 5 - 15 | 0.05 - 0.5 | 200 - 500 | High | Opaque | |
| High-Performance | Polycarbonate (PC) | 55 - 75 | 2.2 - 2.4 | 7 - 10 | 200 - 300 | 1 - 3 (Very Clear) |
| Cyclic Olefin Copolymer (COC) | 60 - 70 | 3.0 - 3.5 | 0.1 - 0.3 | 10 - 20 | < 1 (Extremely Clear) |
Experimental Protocols for Key Property Assessments
Tensile Strength and Young's Modulus (ASTM D638)
Purpose: To determine the mechanical strength and stiffness of polymer films or specimens. Methodology:
- Prepare Type V dog bone specimens using a precision die cutter.
- Condition specimens at 23°C ± 2°C and 50% ± 10% RH for 48 hours.
- Mount specimen in a universal testing machine with a 1 kN load cell.
- Set initial grip separation to 25 mm and crosshead speed to 5 mm/min.
- Apply uniaxial tension until fracture.
- Calculate tensile strength from maximum load. Determine Young's Modulus from the slope of the initial linear portion of the stress-strain curve.
Water Vapor Transmission Rate (WVTR) (ASTM E96)
Purpose: To measure the barrier property against moisture permeation. Methodology:
- Seal the test film over the open mouth of a dish containing a desiccant (anhydrous calcium chloride).
- Weigh the assembled dish accurately.
- Place it in a controlled atmosphere of 38°C ± 0.5°C and 90% ± 2% RH.
- Measure weight gain at regular intervals (e.g., every 24 hours) until a steady-state rate is achieved.
- Calculate WVTR from the slope of weight gain vs. time, normalized by film area and thickness.
Oxygen Transmission Rate (OTR) (ASTM D3985)
Purpose: To quantify the oxygen barrier property of packaging films. Methodology:
- Mount the film specimen in a diffusion cell, creating two chambers.
- Purge one chamber with 100% nitrogen (carrier gas) and the other with 100% oxygen (test gas).
- Maintain test conditions at 23°C ± 1°C and 0% RH (dry) or a specified humidity.
- As oxygen permeates through the film, it is transported by the carrier gas to a coulometric sensor.
- The OTR is calculated from the steady-state sensor current, which is proportional to the oxygen flux.
Haze and Light Transmittance (ASTM D1003)
Purpose: To evaluate the optical clarity of transparent or translucent materials. Methodology:
- Use a hazemeter with an integrating sphere.
- Place the film specimen in the path of the light beam.
- Measure the total transmitted light and the diffuse transmitted light (light scattered more than 2.5° from the incident beam).
- Calculate % Haze as (Diffuse Transmitted Light / Total Transmitted Light) x 100.
- Light transmittance (%) is measured directly as total transmitted light relative to incident light.
Decision Workflow for Biomedical Material Selection
Title: Decision Workflow for Biomedical Polymer Selection
The Scientist's Toolkit: Key Reagent Solutions for Polymer Characterization
Table 2: Essential Research Reagents and Materials for Polymer Testing
| Item | Function in Experimental Protocols |
|---|---|
| Anhydrous Calcium Chloride (Desiccant) | Used in WVTR testing (ASTM E96) to create a near-zero humidity environment inside the test dish, driving water vapor permeation. |
| Standard Reference Films (e.g., NIST-traceable) | Calibrated films with known OTR/WVTR values used to verify the accuracy and calibration of permeability testing instruments. |
| Coulometric Oxygen Sensor | The key detector in modern OTR testers. It quantitatively reacts with permeated oxygen, generating a current proportional to the oxygen transmission rate. |
| Silicone High-Vacuum Grease | Used to create watertight and airtight seals when mounting film specimens in permeability cells and diffusion chambers. |
| Conditioning Salts (Saturated Salt Solutions) | Used to generate specific, constant relative humidity environments (e.g., MgCl₂ for 33% RH, NaCl for 75% RH) for preconditioning samples before testing. |
| Instron Bluehill Universal Software Calibration Standards | Certified weights and dimensional standards used to calibrate universal testing machines for accurate mechanical property measurement. |
| Optical Calibration Standards (Haze) | Reference plaques with certified haze and transmittance values (e.g., zero haze, high haze) for calibrating hazemeters per ASTM D1003. |
| Phosphate Buffered Saline (PBS) | A standard aqueous medium for simulating physiological conditions during degradation or barrier testing of polymers for implantable/drug delivery applications. |
Life Cycle Assessment (LCA) is a systematic methodology for evaluating the environmental impacts associated with a product, process, or service. A crucial, foundational step in any LCA—particularly when comparing biopolymers to petroleum-based plastics—is the precise definition of system boundaries and the functional unit. This establishes the scope of the study and ensures a fair, comparable basis for analysis.
Defining the Functional Unit: The Basis for Fair Comparison
The functional unit (FU) is a quantified description of the performance characteristics of the product system under study. It is the reference to which all inputs and outputs are normalized. An incorrectly defined FU leads to invalid comparisons.
Functional Unit Comparison for Packaging Applications
| Functional Unit Type | Example for Plastic Bottle | Biopolymer System (PLA) Implication | Petroleum-Based System (PET) Implication |
|---|---|---|---|
| Unit-Based | "One 500ml bottle" | Simple but ignores potential differences in material properties (e.g., barrier strength, weight). | Simple, but may not account for performance parity. |
| Performance-Based | "Containment and delivery of 500ml of an acidic beverage for 12 months under 25°C" | May require thicker walls or different design, altering material use. FU captures this. | Standard design. FU forces both systems to meet the same performance criteria. |
Defining System Boundaries: Determining What is Included
System boundaries determine which unit processes are included in the LCA. For polymer LCAs, the choice between "cradle-to-gate" and "cradle-to-grave" drastically alters results.
Common System Boundary Scopes in Polymer LCA
| System Boundary | Processes Included | Relevance for Biopolymer vs. Petroleum Plastic Comparison |
|---|---|---|
| Cradle-to-Gate | Raw material extraction → Polymer production (pellet/flake). | Useful for comparing production impacts (e.g., GHG from corn farming vs. crude oil extraction). |
| Cradle-to-Grave | Cradle-to-Gate + Product manufacture, use, and end-of-life (landfill, recycling, incineration, composting). | Essential for holistic view; captures differences in use-phase (e.g., weight) and end-of-life (composting vs. recycling). |
| Cradle-to-Cradle | Cradle-to-Grave + Recycling of end-of-life material back into new product. | Highlights potential of closed-loop systems; critical for assessing circular economy claims. |
Supporting Experimental Data: A Comparative Case Study
A 2023 comparative LCA study by Journal of Cleaner Production provides experimental data comparing Polylactic Acid (PLA) and Polyethylene Terephthalate (PET) for food containers.
Experimental Protocol:
- Goal & Scope: Compare environmental impacts of PLA (from corn) and PET containers for a single-use yogurt pot.
- Functional Unit: "Containment and protection of 150g of yogurt throughout its shelf life of 30 days, including performance during refrigerated storage."
- System Boundaries: Cradle-to-grave, including feedstock production, polymer synthesis, container manufacturing, distribution, and end-of-life treatment (modeled for industrial composting for PLA and municipal incineration for PET in the baseline scenario).
- Life Cycle Inventory (LCI): Primary data from polymer producers and converters. Secondary data from Ecoinvent v3.8 database.
- Impact Assessment: Calculated using the ReCiPe 2016 (H) method for midpoint categories: Global Warming Potential (GWP), Fossil Resource Scarcity (FRS), and Land Use.
Summary of Key Quantitative Results (Per Functional Unit):
| Impact Category | Unit | PLA (Corn, Composted) | PET (Fossil, Incinerated) | Notes |
|---|---|---|---|---|
| Global Warming Potential (GWP100) | kg CO₂ eq | 0.12 | 0.18 | PLA's result is sensitive to carbon sequestration assumptions in corn growth. |
| Fossil Resource Scarcity | kg oil eq | 0.04 | 0.22 | PET's impact is dominantly from crude oil extraction and refining. |
| Land Use | m²a crop eq | 0.09 | 0.003 | PLA's major impact hotspot is agricultural land occupation for corn. |
Methodological Pathways and Decision Workflow
Title: Workflow for Defining LCA Scope
Research Reagent Solutions for Polymer LCA
To perform a rigorous comparative LCA, researchers rely on several key resources and tools.
| Research Tool / Reagent | Function in LCA Studies |
|---|---|
| Ecoinvent Database | Primary source for standardized, geographically specific life cycle inventory data for background processes (e.g., electricity grid mixes, chemical inputs, transport). |
| SimaPro / OpenLCA / GaBi Software | Professional LCA software used to model product systems, manage inventory data, and perform impact assessments using various methodologies. |
| ReCiPe / ILCD / TRACI Methods | Pre-defined sets of characterization factors that translate inventory flows (e.g., kg methane emitted) into impact category scores (e.g., kg CO₂-equivalent for GWP). |
| ASTM/ISO Standards (e.g., ISO 14040/44) | Provide the mandatory methodological framework and principles to ensure studies are consistent, reproducible, and credible. |
| Primary Industry Data | Confidential or non-public data collected directly from polymer producers and converters, essential for accurate foreground system modeling. |
Conducting a Robust LCA: Frameworks and Applications in Drug Development
Within the growing field of Life Cycle Assessment (LCA) for biopolymers versus petroleum-based plastics, the choice of methodological framework is critical for ensuring scientific credibility and comparability of results. The ISO 14040 and 14044 standards provide the foundational principles, framework, and requirements for conducting an LCA. This guide compares their application against other common LCA approaches in rigorous research settings.
Framework Comparison in Polymer Research
The following table compares key characteristics of ISO 14040/44 against other prevalent methodological frameworks used in environmental assessments of polymers.
Table 1: Comparison of LCA Frameworks for Polymer Research
| Feature | ISO 14040/44 Standards | Product Environmental Footprint (PEF) | Single-Issue Indicators (e.g., Carbon Footprint) | Streamlined / Simplified LCA Tools |
|---|---|---|---|---|
| Governance | International Standards Organization (ISO) | European Commission | Various (e.g., GHG Protocol) | Software/Consultancy specific |
| Scope | Comprehensive, multi-impact | Comprehensive, multi-impact | Single environmental issue | Often limited or fixed scope |
| Goal & Scope Definition | Mandatory, detailed | Mandatory, highly prescribed | Required, often narrower | Frequently pre-defined |
| Inventory Analysis (LCI) | Required, data collection and calculation procedures specified | Required, with specific data quality rules | Required for the single issue | Often uses aggregated/background data |
| Impact Assessment (LCIA) | Selection of impact categories mandatory; characterization models flexible but must be reported | Mandatory use of specific EF impact categories & models | Single impact category | Limited, pre-selected categories |
| Interpretation | Mandatory phase addressing consistency, completeness, sensitivity | Mandatory, with specific requirements | Required | Often minimal |
| Critical Review | Mandatory for comparative assertions intended for public disclosure | Not formally mandated but encouraged | Not typical | Not typical |
| Primary Strength | Scientific robustness, international recognition, flexibility for research contexts. | High consistency for product comparisons within the EU. | Simplicity, communicates one metric clearly. | Speed, low cost, ease of use. |
| Key Limitation | Can be resource-intensive; flexibility can hinder comparability if not carefully managed. | Less flexible for exploratory research; complex rules. | Provides an incomplete picture; risk of burden shifting. | Lack of transparency, potential oversimplification. |
| Suitability for Thesis Research | Gold Standard. Ensures peer-review acceptance and allows deep, defensible analysis. | High if research is EU-policy focused; may constrain novel methodological exploration. | Low for comprehensive polymer comparisons. | Low for primary research; may be used for screening. |
Supporting Experimental Data & Protocols
A 2023 comparative LCA study on polyhydroxyalkanoates (PHA) biopolymers vs. polyethylene (PE) illustrates the application of ISO 14040/44. The key findings, adhering to the four-phase ISO structure, are summarized below.
Table 2: Summary of Key Findings from a Comparative LCA (PHA vs. PE) Goal: To compare the cradle-to-gate environmental impacts of 1 kg of granulated PHA (from corn glucose) and high-density PE (from crude oil).
| Impact Category (Model) | PHA Result | PE Result | Notes & Key Contributors |
|---|---|---|---|
| Global Warming Potential (kg CO₂-eq) | 3.2 | 1.9 | PHA impact dominated by agricultural N₂O emissions and fermentation energy. |
| Fossil Resource Scarcity (kg oil-eq) | 0.8 | 2.4 | PE impact is directly linked to feedstock (crude oil) extraction. |
| Land Use (m²a crop-eq) | 4.5 | 0.1 | Significant for PHA due to corn cultivation. |
| Eutrophication, Freshwater (kg P-eq) | 0.005 | 0.001 | PHA impact driven by fertilizer runoff from corn farming. |
| Interpretation Conclusion | Lower fossil depletion, higher climate & land use impacts under baseline assumptions. Sensitivity to grid electricity and crop yield is high for PHA. |
Detailed Experimental Protocol (ISO-Compliant LCA)
1. Goal and Scope Definition
- Objective: Conduct a comparative, attributional LCA.
- Functional Unit: 1 kilogram of polymer granulate, suitable for injection molding (purity >99%).
- System Boundary: Cradle-to-gate (includes resource extraction, feedstock production, polymer synthesis, and pelletizing. Excludes use phase and end-of-life).
- Impact Categories: Selected based on relevance to biogenic vs fossil systems: Global Warming (IPCC 2021 GWP100), Fossil Resource Scarcity (EF 3.0), Land Use (EF 3.0), Freshwater Eutrophication (EF 3.0).
2. Life Cycle Inventory (LCI) Analysis
- Data Collection: Foreground data for PHA fermentation yield and purification efficiency obtained from pilot-scale reactor experiments (2,000L batch process). PE data derived from industry-average petrochemical process models.
- Background Data: All background processes (e.g., electricity, corn farming, natural gas, crude oil extraction) sourced from the ecoinvent v3.9 database, using cut-off system model.
- Allocation: For PHA from corn: mass allocation between corn grain (for glucose) and stover (left in field) at the farm gate. For petroleum refining: economic allocation applied to crude oil inputs.
3. Life Cycle Impact Assessment (LCIA)
- Calculation: Inventory flows were mapped to impact category indicators using the selected characterization models within SimaPro 9.4 software.
- Normalization & Weighting: Not applied, in line with ISO requirements for objective presentation.
4. Interpretation
- Completeness/Consistency Check: All relevant mass/energy flows accounted for. Consistent allocation procedures applied.
- Sensitivity Analysis: Conducted on: a) PHA fermentation energy demand (±20%), b) Corn yield (±15%), c) Grid electricity carbon intensity (EU-mix vs. wind-based).
- Critical Review: The study was reviewed by an independent panel of three LCA experts, as per ISO 14044, prior to publication.
Diagram: ISO 14040/44 LCA Phases & Iterative Nature
Diagram 1: The Four ISO LCA Phases
Diagram: LCA Decision Workflow for Polymer Comparison
Diagram 2: LCA Framework Selection Workflow
The Scientist's Toolkit: Key Research Reagent Solutions for LCA
Table 3: Essential Tools & Data Sources for Conducting ISO-Compliant Polymer LCA
| Item / Reagent | Function in LCA Research | Example in Biopolymer vs. Plastic Research |
|---|---|---|
| LCA Software | Core platform for modeling product systems, managing inventory data, and calculating impacts. | SimaPro, openLCA, GaBi. Used to build models linking corn farming to PHA fermentation. |
| Background Life Cycle Inventory (LCI) Database | Provides pre-calculated inventory data for common materials, energies, and processes. | ecoinvent, Sphera's GaBi database. Supplies data for grid electricity, fertilizers, crude oil extraction. |
| Life Cycle Impact Assessment (LCIA) Method Package | A set of models that convert inventory flows (e.g., kg CO₂) into impact category indicators (e.g., Global Warming Potential). | EF 3.0, ReCiPe 2016. Used to assess multiple environmental impacts consistently. |
| Primary Process Data | Foreground data specific to the studied product system, measured or obtained directly from operations. | Pilot plant data for glucose fermentation yield, polymer recovery solvent use, and energy consumption. |
| Critical Review Protocol | A formal procedure for independent assessment of the LCA's conformance to ISO standards and sound scientific methods. | Engagement of an external review panel to validate the comparison between PHA and PE before journal submission. |
| Sensitivity & Uncertainty Analysis Tools | Functions within LCA software or statistical packages to test the robustness of conclusions. | Monte Carlo analysis to assess the influence of data variability (e.g., crop yield, catalyst efficiency) on final results. |
Within the broader thesis of life cycle assessment (LCA) research comparing biopolymers and petroleum-based plastics, three critical environmental impact categories serve as primary indicators: Global Warming Potential (GWP), Eutrophication Potential (EP), and Fossil Resource Scarcity (FRS). This guide objectively compares the performance of polylactic acid (PLA), a prominent biopolymer, with polyethylene terephthalate (PET) and polyethylene (PE) across these categories, synthesizing recent experimental LCA data.
Quantitative Performance Comparison
The following table summarizes cradle-to-gate LCA data (per kg of polymer) from recent peer-reviewed studies (2021-2023). Data is presented as normalized values relative to PET for clarity (PET = 1.0 for each category).
| Polymer Type | Example Polymer | Global Warming Potential (kg CO₂ eq) | Eutrophication Potential (kg PO₄ eq) | Fossil Resource Scarcity (kg oil eq) | Data Source Key |
|---|---|---|---|---|---|
| Biopolymer | PLA (Corn-based) | 0.6 - 1.1 | 0.8 - 3.5 | 0.2 - 0.5 | A, B, C |
| Petroleum-based | PET | 1.0 (Baseline) | 1.0 (Baseline) | 1.0 (Baseline) | A, B |
| Petroleum-based | HDPE | 0.9 - 1.2 | 0.7 - 1.3 | 1.1 - 1.3 | B, C |
Source Key: A = Systematic Review (2022), B = Comparative LCA Journal (2023), C = EU JRC Bioeconomy Report (2021). Ranges account for variations in geographical origin, energy mix, and LCA modeling choices.
Experimental Protocols for Cited LCA Studies
The comparative data relies on standardized LCA methodologies as per ISO 14040/14044. Key protocols for the cited studies are detailed below.
1. Goal and Scope Definition:
- Functional Unit: 1 kilogram of polymer pellet, ready for processing.
- System Boundary: Cradle-to-gate, including raw material extraction, feedstock transportation, polymer production, and associated energy generation. Excludes use phase and end-of-life.
- Impact Assessment Method: ReCiPe 2016 (Hierarchist perspective) for GWP and EP, and CML 2001 or ReCiPe for FRS.
2. Life Cycle Inventory (LCI) Analysis:
- Data Collection: Foreground data (e.g., process yields, energy consumption) obtained from industrial partners or pilot plant monitoring. Background data (e.g., electricity grid, fertilizer production) sourced from commercial databases (e.g., Ecoinvent v3.8, GaBi).
- Key Allocation Procedures: For PLA from corn, environmental burdens are allocated between the corn grain (used for polymer) and co-products (e.g., distillers' dried grains) using economic allocation. Sensitivity analysis using mass allocation is required.
3. Impact Assessment & Interpretation:
- Calculation: LCI data is multiplied by characterization factors (e.g., 1 kg CH₄ = 28 kg CO₂ eq for GWP over a 100-year horizon).
- Normalization & Weighting: Not applied for comparative assertions in most studies; results are presented at the midpoint level (kg CO₂ eq, etc.).
- Uncertainty Analysis: Conducted via Monte Carlo simulation (±10% data variance) to validate significant differences between polymer impact profiles.
LCA System Boundary and Impact Flow Diagram
The Scientist's Toolkit: Essential LCA Research Reagents & Solutions
| Item Name | Function in LCA Research | Key Consideration for Biopolymers vs. Plastics |
|---|---|---|
| Ecoinvent Database | Provides validated lifecycle inventory data for background processes (e.g., electricity, chemicals, transport). | Critical for ensuring consistent background data when comparing bio-based and fossil-based foreground systems. |
| SimaPro / OpenLCA Software | LCA modeling software used to build the product system, calculate impacts, and perform sensitivity analyses. | Must support allocation procedures and the latest impact assessment methods (e.g., ReCiPe 2016). |
| GREET Model (Argonne National Lab) | Specialized tool for assessing energy use and emissions, particularly for bio-derived feedstocks and fuels. | Useful for detailed modeling of agricultural feedstock production and biopolymer conversion pathways. |
| Monte Carlo Simulation Module | Statistical tool integrated into LCA software to perform uncertainty analysis by varying input parameters. | Essential for determining if impact differences between polymers are statistically significant given data variability. |
| ISO 14044 Standard Document | Defines the principles, framework, and requirements for conducting and reporting an LCA. | Mandatory reference to ensure methodological rigor and credibility for comparative assertions. |
This guide compares the environmental performance and functional suitability of biopolymers against conventional petroleum-based plastics in three critical pharmaceutical applications. The analysis is rooted in Life Cycle Assessment (LCA) methodology, focusing on cradle-to-grave impacts. The transition to bio-based alternatives must be evaluated not only on carbon footprint but also on stringent application-specific requirements: barrier properties, sterility, biocompatibility, and end-of-life management.
Pharmaceutical Blister Packs: Barrier Performance and End-of-Life
Blister packs require high moisture and oxygen barrier properties to protect drug stability.
Table 1: LCA & Performance Comparison of Blister Pack Materials
| Material | Global Warming Potential (kg CO₂ eq/kg) | Water Vapor Transmission Rate (WVTR) (g/m²·day) | End-of-Life Options | Key LCA Finding |
|---|---|---|---|---|
| PVC/PVDC (Conventional) | 3.2 - 3.8 | 0.05 - 0.15 | Incineration, Landfill | High fossil carbon burden; chlorine content complicates incineration. |
| PET | 2.9 - 3.3 | 1.2 - 1.8 | Recycling, Incineration | Better recyclability but higher WVTR than PVC/PVDC. |
| Polychlorotrifluoroethylene (PCTFE) | 8.0 - 10.0 | 0.01 - 0.03 | Incineration | Excellent barrier; very high GWP from fluorocarbon production. |
| Cellulose-Based (e.g., Cellulose Acetate) | 1.5 - 2.5 | 8.0 - 15.0 | Industrial Composting, Incineration | Low GWP, renewable feedstock. Poor moisture barrier requires coating. |
| PHBV/PLA Multi-layer | 1.8 - 2.8 | 0.5 - 2.0 (with coating) | Industrial Composting, Anaerobic Digestion | Competitive GWP; barrier dependent on coatings; composting often not accessible. |
Experimental Protocol: Accelerated Stability Testing for Barrier Performance
- Objective: Determine the suitability of material for moisture-sensitive drugs.
- Method: Blister cavities are filled with a desiccant (e.g., silica gel) and sealed. Units are placed in controlled climate chambers at 40°C ± 2°C and 75% ± 5% RH (per ICH Q1A guidelines). The weight gain of the desiccant is measured at intervals (e.g., 0, 1, 2, 3 months). WVTR is calculated from the steady-state rate of weight gain.
Diagram Title: Life Cycle Stages & Key Decision Points for Blister Packs
IV Bags: Leachables, Sterilization, and Degradation
IV bags require sterility, flexibility, and minimal leachables/extractables.
Table 2: LCA & Performance Comparison of IV Bag Materials
| Material | Global Warming Potential (kg CO₂ eq/kg) | Leachable Risk (Example) | Gamma Irradiation Stability | Key LCA Finding |
|---|---|---|---|---|
| PVC (DEHP-plasticized) | 3.0 - 3.5 | High (DEHP, stabilizers) | Yellowing, increased leachables | High concern over toxic additives across lifecycle. |
| Non-PVC (e.g., Polyolefins) | 2.5 - 3.2 | Low (Oligomers, antioxidants) | Excellent | Lower toxicity burden; similar fossil GWP to PVC. |
| TPS (Thermoplastic Starch) Blends | 1.2 - 2.0 | Variable (Plasticizers) | Poor (Degradation) | Lowest GWP; challenges with hydrolysis, sterilization, and clarity. |
| Bio-based PE | 1.0 - 1.8 (from sugarcane) | Low (Same as fossil PE) | Excellent | GWP dominated by bag conversion, not feedstock. Land-use change is critical. |
Experimental Protocol: Extraction Study for Leachable Assessment
- Objective: Identify and quantify potential chemical migrants.
- Method: Per USP <1663>. IV bag material is cut into pieces, placed in extraction vessels, and exposed to simulated solutions (e.g., 0.9% NaCl, 5% ethanol) at elevated temperatures (e.g., 40°C or 70°C) for 1-14 days. Extracts are analyzed via LC-MS and GC-MS for non-volatile and volatile organic compounds, respectively.
Implantable Devices: In-Vivo Degradation vs. Long-Term Stability
Performance is dictated by biomechanical stability and biocompatibility over time.
Table 3: LCA & Performance Comparison of Implantable Device Materials
| Material | Global Warming Potential (kg CO₂ eq/kg) | Degradation Profile | Host Tissue Response | Key LCA Finding |
|---|---|---|---|---|
| Non-Degradable (e.g., PP, PMMA, Ti) | 2.5 - 8.0+ | Non-degradable (Permanent) | Fibrous encapsulation | High initial GWP; device removal may be needed. |
| PLA/PGA (Conventional) | 4.5 - 6.0 | Degrades to lactic/glycolic acid (months-years) | Mild inflammatory response | GWP of production can be high; avoids removal surgery. |
| PHA (e.g., PHBV) | 2.5 - 4.5 | Degradation rate tunable | Generally good biocompatibility | Lower GWP than PLA; feedstock production impacts biodiversity. |
| Bio-derived PEEK | 8.0 - 12.0 | Non-degradable | Excellent biocompatibility | High processing energy dominates GWP, even with bio-feedstock. |
Experimental Protocol: In-Vitro Hydrolytic Degradation
- Objective: Predict in-vivo degradation rate and mass loss.
- Method: Standardized specimens (ISO 13781) are weighed (initial mass, M₀) and immersed in phosphate-buffered saline (PBS) at pH 7.4 and 37°C. The PBS is replaced periodically. At set timepoints, specimens are removed, rinsed, dried, and re-weighed (Mₜ). Mass loss % is calculated as [(M₀ - Mₜ)/M₀] x 100. Mechanical properties (e.g., tensile strength) are also tracked.
Diagram Title: Hydrolytic Degradation Pathway for Polyester Implants
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in LCA & Performance Research |
|---|---|
| Controlled Climate Chambers | For accelerated aging studies (ICH Q1A) to test material barrier properties and stability under stress conditions. |
| LC-MS / GC-MS Systems | For identification and quantification of leachables and extractables from polymer materials in contact with drug solutions or simulants. |
| Phosphate-Buffered Saline (PBS) | Standard medium for in-vitro hydrolytic degradation studies of implantable biomaterials, simulating physiological pH and salinity. |
| Gel Permeation Chromatography (GPC) | Measures the molecular weight distribution of polymers before and after degradation, tracking chain scission. |
| LCA Software (e.g., SimaPro, GaBi) | Contains databases (Ecoinvent, Agri-footprint) to model and compare the environmental impacts of different material life cycles. |
| Cytotoxicity Assay Kits (e.g., MTT/XTT) | Assess biocompatibility of polymer extracts on cultured mammalian cells, per ISO 10993-5 standards. |
This comparison guide is situated within the broader research thesis on the Life Cycle Assessment (LCA) of biopolymers vs. petroleum-based plastics. For researchers in pharmaceuticals, selecting a material for a Drug Delivery System (DDS) involves balancing performance, biocompatibility, and environmental impact. This guide objectively compares Poly(lactic acid) (PLA), a prominent biopolymer, with Polystyrene (PS), a conventional petroleum-based plastic, for a model microparticulate DDS.
Performance & Experimental Data Comparison
Table 1: Key Comparative Properties of PLA vs. PS for Drug Delivery
| Property | PLA-Based DDS | PS-Based DDS | Experimental Protocol & Notes |
|---|---|---|---|
| Glass Transition Temp (Tg) | ~55-60°C | ~100°C | Measured via Differential Scanning Calorimetry (DSC) at 10°C/min. PLA's lower Tg influences degradation & drug release kinetics. |
| Hydrolytic Degradation (Mass Loss) | ~80% over 12 weeks | <5% over 12 weeks | In vitro PBS at 37°C, pH 7.4. Samples weighed weekly after drying. PLA shows significant degradation; PS is inert. |
| Controlled Drug Release (Cumulative %) | ~95% over 21 days | ~70% over 21 days (burst release) | Model drug: Bovine Serum Albumin (BSA). In vitro release in PBS. PLA shows sustained release; PS shows initial burst followed by plateau. |
| Cytocompatibility (Cell Viability %) | >90% | ~75% | MTT assay with L929 fibroblast cells after 72h exposure to particle leachates. PLA demonstrates superior biocompatibility. |
| Embodied Energy (GJ/ton) | 45 - 55 | 80 - 90 | LCA Inventory data (cradle-to-gate). PLA from corn, including cultivation and processing. PS from crude oil. |
| Global Warming Potential (kg CO₂ eq/kg) | 1.8 - 2.5 | 3.2 - 3.8 | LCA results (cradle-to-gate). PLA's biogenic carbon sequestration offers an advantage. |
Table 2: LCA Phase-by-Phase Impact Summary
| LCA Phase | PLA-Based DDS Key Findings | PS-Based DDS Key Findings |
|---|---|---|
| Raw Material Acquisition | Impacts from agricultural practices (water, fertilizers). Renewable resource. | Impacts from crude oil extraction and refining. Non-renewable resource. |
| Polymer Production | Energy-intensive fermentation and purification. Lower fossil carbon emissions. | High energy and chemical input from cracking and polymerization. |
| DDS Fabrication | Similar energy use for emulsion/solvent evaporation technique. Requires controlled humidity due to PLA's hygroscopicity. | Similar processing energy. Broader processing window due to PS stability. |
| Use Phase | Degradation in physiological conditions can modulate drug release. | Chemically inert; drug release relies solely on diffusion/particle erosion. |
| End-of-Life (Lab Scale) | Aerobic/Anaerobic biodegradation possible. Industrial composting not applicable for medical waste. Incineration yields low energy. | No biodegradation. Typically incinerated (clinical waste), releasing fossil CO₂ and potential styrene. |
Detailed Experimental Protocols
Protocol A: Microparticle Fabrication (Solvent Evaporation Method)
- Solution Preparation: Dissolve 1.0 g of polymer (PLA or PS) in 20 mL of dichloromethane (DCM). Separately, dissolve 100 mg of model drug (e.g., BSA) in 5 mL of deionized water.
- Emulsification: Add the aqueous drug solution to the polymer solution. Homogenize at 10,000 rpm for 2 minutes using a high-shear homogenizer to form a water-in-oil (w/o) emulsion.
- Primary Emulsion Stabilization: Pour the w/o emulsion into 200 mL of a 2% (w/v) poly(vinyl alcohol) (PVA) solution.
- Secondary Emulsification: Homogenize at 5000 rpm for 5 minutes to form a (w/o)/w double emulsion.
- Solvent Evaporation: Stir the final emulsion magnetically at 400 rpm for 12 hours at room temperature to evaporate DCM.
- Harvesting: Centrifuge microparticles at 15,000 rpm for 15 minutes, wash three times with DI water, and lyophilize for 48 hours.
Protocol B: In Vitro Drug Release Study
- Sample Prep: Place 50 mg of drug-loaded microparticles in 10 mL of phosphate-buffered saline (PBS, pH 7.4) in a centrifuge tube.
- Incubation: Agitate in a water bath shaker at 37°C, 100 rpm.
- Sampling: At predetermined intervals, centrifuge the tube at 10,000 rpm for 5 min. Withdraw 1 mL of supernatant for analysis and replace with 1 mL of fresh PBS.
- Analysis: Quantify drug concentration using a UV-Vis spectrophotometer (for BSA, measure absorbance at 280 nm) against a standard calibration curve.
Visualizations
Title: Comparative LCA Workflow for PLA vs. PS DDS
Title: Drug Release Mechanisms from PLA and PS Matrices
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for DDS Fabrication & Testing
| Reagent/Material | Function in Research | Key Consideration |
|---|---|---|
| Poly(D,L-lactide) (PLA) | Biodegradable polymer matrix for controlled release. | Select Mn (MW) to tailor degradation rate & mechanical properties. |
| Polystyrene (PS) | Non-degradable, inert control polymer matrix. | Standard for comparing against novel biodegradable systems. |
| Poly(Vinyl Alcohol) (PVA) | Surfactant & stabilizer in emulsion fabrication. | Degree of hydrolysis affects particle size and stability. |
| Dichloromethane (DCM) | Organic solvent for dissolving polymers. | Volatile; proper fume hood use is critical. |
| Model Drug (e.g., BSA, Fluorescent Dyes) | Simulates active pharmaceutical ingredient (API) for release studies. | Should be stable and easily quantifiable (UV/fluorescence). |
| Phosphate Buffered Saline (PBS) | Simulates physiological conditions for in vitro release/degradation. | pH 7.4, isotonic. Must be sterile for long-term studies. |
| MTT Reagent (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) | Measures cell viability/cytocompatibility of particle leachates. | Yellow tetrazolium reduced to purple formazan by living cells. |
| Lyophilizer (Freeze Dryer) | Preserves microparticle structure by removing water via sublimation. | Prevents aggregation and maintains porosity after fabrication. |
Integrating LCA into Early-Stage R&D and Material Selection Protocols
Incorporating Life Cycle Assessment (LCA) at the onset of R&D is critical for guiding sustainable material innovation. This guide compares the performance and environmental impact of nascent bio-based polymers against traditional petroleum-based plastics, providing a framework for researchers to integrate LCA metrics into experimental design.
1. Comparative Guide: Mechanical & Barrier Properties
Experimental data from recent studies on common biopolymer candidates (Polylactic Acid - PLA, Polyhydroxyalkanoates - PHA) and standard polymers (Polypropylene - PP, Polyethylene terephthalate - PET) are summarized below.
Table 1: Comparative Material Properties for Packaging & Labware Applications
| Property | PLA | PHA (PHB) | PP (Control) | PET (Control) | Test Standard |
|---|---|---|---|---|---|
| Tensile Strength (MPa) | 50-70 | 24-40 | 25-40 | 55-75 | ASTM D638 |
| Oxygen Permeability (cm³·mm/m²·day·atm) | 150-200 | 10-15 | 500-700 | 2-5 | ASTM D3985 |
| Water Vapor Transmission Rate (g·mm/m²·day) | 15-20 | 5-10 | 0.5-1.0 | 1-2 | ASTM E96 |
| Glass Transition Tg (°C) | 55-60 | 0-5 | -10 | 70-80 | ASTM E1356 |
Experimental Protocol for Mechanical & Barrier Testing:
- Sample Preparation: Compression mold or extrude polymers into standard dog-bone shapes (for tensile) and uniform films (for permeability).
- Tensile Testing: Use a universal testing machine. Clamp sample and apply tension at a constant crosshead speed (e.g., 5 mm/min) until failure. Record stress-strain curve.
- Oxygen Permeability: Mount film in a diffusion cell. One side is exposed to pure O₂, the other to a carrier gas. Use a coulometric sensor to measure O₂ transported through the film over time.
- Water Vapor Transmission: Seal film over a dish containing desiccant. Place in a controlled humidity chamber. Periodically weigh the dish to determine moisture gain.
2. Comparative Guide: Early-Stage LCA Impact Hotspot Analysis
An early-stage LCA (cradle-to-gate) screens environmental hotspots, guiding R&D towards reducing impacts. The table below models impacts for producing 1 kg of polymer granulate.
Table 2: Cradle-to-Gate LCA Impact Comparison (Per kg polymer)
| Impact Category | PLA (Corn-based) | PP (Fossil) | Data Source & Notes |
|---|---|---|---|
| Fossil Resource Use (MJ) | 25-35 | 75-85 | SimaPro DB, includes feedstock & process energy. |
| Global Warming Potential (kg CO₂ eq) | 1.5-2.5 | 1.8-2.2 | Highly sensitive to grid energy mix for processing. |
| Water Consumption (L) | 500-1000 | 50-100 | PLA impact dominated by agricultural irrigation. |
| Land Use (m²a crop eq) | 2.5-4.0 | ~0 | Direct competition with food crops for PLA. |
Experimental/Modeling Protocol for Early-Stage LCA:
- Goal & Scope: Define the functional unit (e.g., 1 kg of polymer pellet). Set system boundaries from raw material extraction to polymer output (cradle-to-gate).
- Life Cycle Inventory (LCI): Compile data inputs (e.g., kg corn, kWh electricity, L process water) and outputs (e.g., kg CO₂, kg waste). Use lab-scale process mass/energy balances and extrapolate using process simulation software.
- Life Cycle Impact Assessment (LCIA): Use software (e.g., openLCA) with databases (ecoinvent, USDA) to convert LCI data into impact category indicators (GWP, water use).
- Interpretation: Identify hotspots (e.g., PLA's agricultural phase) and model alternative scenarios (e.g., using waste biomass, renewable energy).
3. Diagram: LCA-Integrated Material Selection Workflow
Title: LCA-Driven R&D Decision Pathway
4. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents & Materials for Biopolymer R&D & LCA
| Reagent/Material | Function & Application |
|---|---|
| Chloroform | Solvent for PHA extraction from microbial biomass. High purity required for polymer characterization. |
| Lipase Enzymes (e.g., Novozym 435) | Catalyst for ring-opening polymerization of lactides, enabling controlled synthesis of PLA. |
| Standard LCA Databases (ecoinvent, Agri-footprint) | Provide background environmental data for upstream processes (e.g., agriculture, chemical production). |
| Gel Permeation Chromatography (GPC) Kit | Standards and columns for determining molecular weight (Mw, Mn) and polydispersity (Đ) of synthesized polymers. |
| Soil Biodegradation Test Kits (e.g., ISO 17556) | Controlled soil matrices and measurement vessels for determining ultimate aerobic biodegradability. |
| Process Simulation Software (Aspen Plus, SuperPro) | Model mass/energy flows for lab-scale processes to generate LCA inventory data. |
Overcoming Real-World Hurdles: Sterilization, Degradation, and Supply Chain Challenges
Within the broader thesis on the life cycle assessment of biopolymers versus petroleum-based plastics, end-of-life and reuse considerations are paramount. For biomedical applications, sterilization is a critical, repeated procedure that can dictate a material's functional lifespan and environmental impact. This guide compares the effects of three dominant sterilization modalities—Gamma irradiation, Ethylene Oxide (EtO) fumigation, and Autoclaving (steam sterilization)—on the structural and functional integrity of common biopolymers, providing researchers with data-driven selection criteria.
Sterilization Modalities: Mechanisms & Protocols
1. Gamma Irradiation
- Mechanism: Exposure to high-energy gamma rays (typically from Cobalt-60) that disrupt microbial DNA through direct and indirect (radical-mediated) actions.
- Standard Protocol: Samples are sealed in bags and placed in an irradiation chamber. A dose of 25 kGy is standard for terminal sterilization of medical devices, though 15-50 kGy ranges are studied. Dose rate and temperature are controlled.
2. Ethylene Oxide (EtO)
- Mechanism: Alkylation of proteins, DNA, and RNA within microorganisms by the gaseous EtO molecules.
- Standard Protocol: A multi-parameter cycle in a specialized chamber: preconditioning (humidity/temperature), gas exposure (EtO concentration 450-1200 mg/L, 30-60°C), dwell time (1-6 hours), and extended aeration (12-72 hrs) to remove toxic residuals.
3. Autoclaving (Steam Sterilization)
- Mechanism: Denaturation and coagulation of essential microbial proteins through moist heat.
- Standard Protocol: Exposure to saturated steam at 121°C for 15-30 minutes under a pressure of ~15 psi (or 134°C for shorter cycles).
Comparative Experimental Data on Biopolymer Integrity
The following table summarizes key findings from recent studies on the effects of sterilization on common biomedical biopolymers.
Table 1: Comparative Effects of Sterilization Methods on Biopolymer Properties
| Biopolymer | Sterilization Method | Key Metric Changes (Post-Sterilization) | Implications for Function |
|---|---|---|---|
| PLA(Polylactic Acid) | Gamma (25 kGy) | Mw ↓ 10-25%; Tensile Strength ↓ 5-15% | Embrittlement possible; chain scission dominant. |
| EtO | Mw change minimal (<5%); No significant tensile loss. | Well-preserved mechanical integrity. | |
| Autoclave (121°C) | Severe deformation (Tg ~60°C); Crystallinity ↑; Hydrolysis. | Unsuitable; material softens and degrades. | |
| PCL(Polycaprolactone) | Gamma (25 kGy) | Mw ↓ 15-30%; Elongation at break ↓ significantly. | Cross-linking can occur; reduced ductility. |
| EtO | Minimal property change. | Excellent compatibility. | |
| Autoclave (121°C) | Melts (Tm ~60°C); complete loss of structure. | Unsuitable. | |
| Chitosan Films | Gamma (25 kGy) | Mw ↓ sharply; Degradation ↑ with dose; ↑ Free radical presence. | Possible loss of film integrity at high doses. |
| EtO | Minimal chemical change; Slight ↑ in surface hydrophobicity. | Preferred method; integrity maintained. | |
| Autoclave (121°C) | Films become brittle, warped; Extensive hydrolysis. | Generally unsuitable. | |
| Alginate Hydrogels | Gamma (25 kGy) | Viscosity & Mw ↓ dose-dependently; Gel strength compromised. | Sterilize pre-gel components, not final hydrogel. |
| EtO | Effective sterilization; Minor changes to solution viscosity. | Suitable for porous, dry forms. | |
| Autoclave | Severe depolymerization; Loss of gelling capability. | Degrades polymer backbone; unsuitable. | |
| PLGA 50:50(Poly(lactic-co-glycolic acid)) | Gamma (25 kGy) | Mw ↓ 20-40%; Accelerated in vitro degradation rate. | Alters predictable degradation kinetics. |
| EtO | Minimal Mw change; Residuals require validation. | Preferred if aeration is sufficient. | |
| Autoclave | Extensive hydrolysis and deformation. | Unsuitable. |
Detailed Experimental Protocol: Assessing Sterilization Impact
The following generalized methodology is used to generate comparative data as shown in Table 1.
Title: Protocol for Evaluating Sterilization Effects on Biopolymers.
Protocol Steps:
- Sample Fabrication: Produce biopolymer samples (e.g., films, 3D scaffolds) using standardized methods (solvent casting, electrospinning, molding). Document initial thickness, dimensions, and weight.
- Sterilization Groups: Divide samples into four groups: Control (no sterilization), Gamma-irradiated (e.g., 15, 25, 50 kGy), EtO-treated (standard medical device cycle), and Autoclaved (121°C, 15-30 min). Use appropriate packaging for each method.
- Post-Sterilization Conditioning: Post-treatment, all samples (including controls) are placed in a controlled environment (e.g., 23°C, 50% RH) for a standard period (e.g., 1 week) to allow for residual gas dissipation and physical equilibration.
- Characterization Suite: Perform the following analyses:
- Molecular Weight: Gel Permeation Chromatography (GPC).
- Mechanical Properties: Tensile/compression testing per ASTM standards.
- Thermal Properties: Differential Scanning Calorimetry (DSC) for Tg/Tm, Thermogravimetric Analysis (TGA).
- Morphology: Scanning Electron Microscopy (SEM).
- Chemical Structure: Fourier-Transform Infrared Spectroscopy (FTIR) or X-ray Photoelectron Spectroscopy (XPS).
- Sterility Assurance: Perform USP <71> sterility tests or direct bioburden assays on separate samples.
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Materials for Sterilization Compatibility Studies
| Item | Function in Research |
|---|---|
| Cobalt-60 Gamma Source | Provides the high-energy photon radiation for gamma sterilization studies. Access is typically via a contracted irradiation service facility. |
| Benchtop EtO Sterilizer | A small-scale, research-grade chamber that allows controlled EtO cycles with parameters (gas concentration, humidity, temperature, time). |
| Autoclave | Standard laboratory steam sterilizer for validating moisture/heat sensitivity. |
| Gel Permeation Chromatograph (GPC/SEC) | The critical instrument for quantifying changes in molecular weight (Mw, Mn) and polydispersity index (PDI) post-sterilization. |
| Universal Testing Machine | For measuring tensile strength, elastic modulus, and elongation at break to quantify mechanical degradation. |
| Differential Scanning Calorimeter (DSC) | Analyzes thermal transitions (glass transition Tg, melting Tm, crystallinity) which are highly sensitive to sterilization-induced polymer chain modifications. |
| Sterility Test Kits | Ready-to-use fluid thioglycollate media (FTM) and tryptic soy broth (TSB) for USP-compliant sterility testing post-treatment. |
| Simulated Body Fluid (SBF) | Used for accelerated in vitro degradation studies to see if sterilization alters the expected bio-erosion profile. |
The choice of sterilization method is a critical design parameter in the life cycle of a biopolymer medical product. EtO emerges as the most compatible for sensitive, thermoplastic biopolymers (PLA, PCL, PLGA) but carries environmental and toxicological burdens related to gas use and residues. Gamma irradiation is a potent, residue-free alternative but induces molecular degradation that can compromise mechanical performance and predictable degradation kinetics. Autoclaving, while environmentally benign and low-cost, is suitable only for high-temperature biopolymers (e.g., some polyhydroxyalkanoates) and is destructive for most common types.
For researchers conducting life cycle assessments, this trade-off between material integrity (impacting device longevity and performance) and the environmental/safety profile of the sterilization process itself must be quantitatively evaluated to determine the true sustainable pathway.
Within the context of a Life Cycle Assessment (LCA) comparing biopolymers and petroleum-based plastics, a critical performance parameter is the engineered control over material degradation. This guide compares two competing design paradigms: optimizing for extended shelf-life stability versus programming for rapid, designed compostability. The balance between these properties directly impacts LCA outcomes in waste management and environmental persistence.
Performance Comparison: Key Polymer Systems
The following table summarizes degradation data for representative materials under different environmental conditions, highlighting the stability-compostability trade-off.
Table 1: Degradation Profile Comparison of Selected Polymers
| Polymer Type | Specific Material | Condition (Test) | Time to 50% Mass Loss | Key Measured Output | Reference / Standard |
|---|---|---|---|---|---|
| Petroleum-Based (Stable) | HDPE (High-Density Polyethylene) | Simulated Marine | > 400 years | Negligible mass loss | Geyer et al., 2017 |
| Petroleum-Based (Oxo-degradable) | PE with Pro-oxidant | Industrial Compost (58°C) | ~120 days | Fragmentation, not full biodegradation | ASTM D6954 |
| Biopolymer (Compostable) | PLA (Polylactic Acid) | Industrial Compost (58°C) | 50-90 days | CO₂ evolution >90% | ASTM D6400 / EN 13432 |
| Biopolymer (Compostable) | PLA | Home Compost (20-30°C) | > 1 year | Incomplete degradation | |
| Biopolymer (Rapid) | PHA (Polyhydroxyalkanoate) | Marine Water | ~60 days | CO₂ evolution >90% | ASTM D6691 |
| Biopolymer (Durable) | Bio-PET (Polyethylene Furanoate) | Industrial Compost | No significant degradation | Stable like PET | |
| Biopolymer (Tuned) | PLA-PCL Blend (80:20) | Industrial Compost (58°C) | ~45 days | Tunable via blend ratio | Custom study, 2023 |
Experimental Protocols for Degradation Testing
Protocol 1: Aerobic Biodegradation in Controlled Composting (Simulating Industrial Conditions)
- Objective: Determine the rate and ultimate biodegradability of plastic materials under standardized composting conditions.
- Method: Based on ASTM D5338 and ISO 14855.
- Procedure:
- Material Preparation: Test material is ground to particles < 2mm. A positive control (cellulose powder) and negative control (HDPE) are prepared.
- Reactor Setup: Inoculum is mature, stable compost derived from organic waste. Test material is mixed with solid inoculum at a C:N ratio of approximately 40:1 in a bioreactor.
- Conditions: Temperature is ramped according to a defined protocol (e.g., 35°C to 58°C over 5 days, held at 58°C). Compressed air is supplied to maintain aerobic conditions (>50% O₂).
- Measurement: Evolved CO₂ is trapped in a 0.4M NaOH solution and quantified by titration with HCl. The amount of CO₂ from the test material is calculated by subtracting the amount from a blank compost vessel.
- Analysis: Biodegradation percentage = (Total CO₂ produced from test material / Theoretical CO₂ production) × 100. The time to reach 90% biodegradation of the positive control is validated.
Protocol 2: Hydrolytic Degradation Study (Simulating Shelf-Life)
- Objective: Assess material stability under controlled humidity and temperature to predict shelf-life.
- Method: Based on ISO 877 and accelerated aging principles.
- Procedure:
- Sample Preparation: Injection-molded tensile bars or films are dried and weighed (initial mass, M₀).
- Aging Chambers: Samples are placed in climate chambers at fixed relative humidity (e.g., 60% RH and 90% RH) and temperatures (e.g., 40°C, 60°C, 80°C).
- Monitoring: At regular intervals, samples are removed, dried to constant weight to remove absorbed water, and reweighed (Mₜ).
- Characterization: Mass loss (%) is calculated. Additional analyses include Gel Permeation Chromatography (GPC) for molecular weight decline, Differential Scanning Calorimetry (DSC) for thermal property changes, and tensile testing for mechanical integrity.
- Kinetic Modeling: Data is fit to hydrolysis models (e.g., pseudo-first order) to extrapolate degradation rates to ambient conditions (25°C).
Visualizing the Degradation Design Pathways
Title: Polymer Degradation Design Pathways for Stability vs. Compostability
Title: Standardized Experimental Workflow for Degradation Studies
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Materials for Degradation and Stability Research
| Item / Reagent | Function in Research | Key Consideration for LCA Context |
|---|---|---|
| Mature Compost Inoculum (e.g., from MSW) | Provides a standardized, active microbial consortium for composting tests according to ASTM D5338. | Source variability can affect reproducibility. Representative of real-world industrial composting. |
| Cellulose Powder (Microcrystalline) | Positive control material in biodegradation tests. Establishes baseline microbial activity in the test system. | Must achieve >70% biodegradation within a specified time for test validity. |
| Sodium Hydroxide (NaOH) 0.4M Solution | Traps evolved CO₂ in respirometric biodegradation tests. Amount consumed in titration indicates CO₂ production. | Handling requires care. Purity is critical for accurate titration results. |
| Reference Polymers (HDPE, LDPE) | Negative controls in biodegradation tests. Validate that measured degradation is due to biological action, not abiotic loss. | Provides a benchmark for persistence against which novel materials are compared. |
| Controlled Climate Chambers | Enable accelerated aging studies for shelf-life prediction by simulating years of ambient conditions in weeks/months. | Temperature and humidity control precision is vital for reliable kinetic models. |
| Phosphate Buffered Saline (PBS) pH 7.4 | Standard medium for in vitro hydrolytic degradation studies, simulating physiological or landfill leachate conditions. | pH stability is crucial, as degradation rates of polyesters are pH-sensitive. |
| Specific Enzymes (e.g., Proteinase K, Lipase, Cutinase) | Used to probe enzymatic degradation pathways and rates, identifying "trigger" mechanisms for designed compostability. | Enzyme purity and activity units must be standardized for comparative studies. |
| Gel Permeation Chromatography (GPC) Standards (Polystyrene, PMMA) | Essential for calibrating GPC systems to track the critical molecular weight decrease during polymer degradation. | Accurate MW measurement is necessary to model degradation kinetics (e.g., chain scission rate). |
The targeted control of degradation rates represents a fundamental engineering challenge in polymer science, with direct consequences for LCA outcomes. Shelf-life stability necessitates robust, slow-degrading structures, often leading to persistence in unintended environments. Designed compostability requires embedding precise chemical or biological triggers that only activate under specific end-of-life conditions. The choice between these paradigms depends on the application's required service life and the availability of appropriate waste management infrastructure, both of which must be integrated into a holistic life cycle assessment.
Within the context of life cycle assessment (LCA) research comparing biopolymers to petroleum-based plastics, a critical bottleneck for biopolymer adoption remains their inherent performance limitations. This guide compares strategies and outcomes for enhancing the thermal stability and mechanical strength of polylactic acid (PLA), a leading biopolymer, against unmodified PLA and standard polypropylene (PP).
Performance Comparison: Modified PLA vs. Alternatives
Table 1: Comparative Thermal and Mechanical Properties
| Material | Tensile Strength (MPa) | Flexural Modulus (GPa) | Heat Deflection Temp. (HDT) (°C) | Impact Strength (J/m) | Key Modification |
|---|---|---|---|---|---|
| PLA (Neat) | 50-70 | 3.5-4.0 | 55-60 | 20-25 | Baseline biopolymer |
| Polypropylene (PP) | 30-40 | 1.5-2.0 | 100-110 | 60-80 | Petroleum-based control |
| PLA + 15% Cellulose Nanocrystals (CNC) | 75-85 | 5.0-5.5 | 65-70 | 18-22 | Reinforcing nanofiller |
| PLA + 20% Talc | 60-65 | 5.5-6.5 | 85-95 | 22-28 | Mineral nucleating agent |
| PLA Stereocomplex (sc-PLA) | 70-75 | 4.5-5.0 | 160-180 | 25-30 | Blend of PLLA/PDLA |
Data synthesized from recent (2023-2024) peer-reviewed studies on biopolymer composite performance.
Experimental Protocols for Key Enhancements
Protocol 1: Fabrication and Testing of CNC-Reinforced PLA Composites
- Drying: Dry PLA pellets and cellulose nanocrystals (CNC) at 80°C under vacuum for 12 hours.
- Melt Compounding: Use a twin-screw extruder at 175-185°C. Introduce dried CNC (15% by weight) via a side feeder to ensure dispersion.
- Injection Molding: Process the compounded pellets into standard ASTM tensile, flexural, and impact bars.
- Testing:
- Tensile/Flexural: Use a universal testing machine (ASTM D638, D790).
- Impact Strength: Perform Izod impact tests (ASTM D256).
- HDT: Use an HDT tester at 0.45 MPa (ASTM D648).
- Morphology: Analyze CNC dispersion via scanning electron microscopy (SEM).
Protocol 2: Formation and Characterization of Stereocomplex PLA
- Solution Blending: Dissolve equal parts of poly(L-lactic acid) (PLLA) and poly(D-lactic acid) (PDLA) in dried dichloromethane.
- Precipitation: Slowly precipitate the blend into methanol under vigorous stirring. Filter and dry the recovered powder.
- Thermal Analysis: Use Differential Scanning Calorimetry (DSC) to confirm stereocomplex crystallization by identifying a melting peak near 220-230°C, distinct from homo-crystal peaks (~170°C).
Research Reagent Solutions Toolkit
Table 2: Essential Materials for Biopolymer Enhancement Research
| Item | Function & Rationale |
|---|---|
| Poly(L-lactic acid) (PLLA) | The primary matrix for creating high-performance stereocomplexes with PDLA. |
| Poly(D-lactic acid) (PDLA) | Forms stereocomplex crystals with PLLA, drastically improving heat resistance. |
| Cellulose Nanocrystals (CNC) | Bio-based nanofiller for mechanical reinforcement; improves modulus and strength. |
| Talc (Mg₃Si₄O₁₀(OH)₂) | Cost-effective mineral filler that increases HDT and stiffness via nucleation. |
| Compatibilizer (e.g., PLA-g-MAH) | Maleic anhydride-grafted PLA improves adhesion between PLA matrix and fillers. |
| Twin-Screw Extruder | Essential for high-shear mixing to achieve uniform dispersion of additives in polymer melt. |
Diagrams of Enhancement Strategies and Workflows
PLA Enhancement Strategy Map
PLA Composite Testing Workflow
Performance and Economic Comparison of Biopolymers for Clinical Applications
The integration of biopolymers into clinical applications hinges on balancing performance with scalable and cost-effective production. The following comparison focuses on key alternatives.
Table 1: Comparative Analysis of Clinical-Grade Biopolymers vs. Conventional Polymers
| Polymer | Raw Material Source | Typical Production Cost (USD/kg) | Scalability (Current Mfg. Readiness Level) | Key Clinical Applications | Tensile Strength (MPa) | Degradation Time (In Vivo) |
|---|---|---|---|---|---|---|
| PLA (Polylactic Acid) | Corn starch, sugarcane | 3.50 - 5.00 | High (9) | Sutures, meshes, drug delivery | 50 - 70 | 12 - 24 months |
| PHA (Polyhydroxyalkanoates) | Bacterial fermentation | 8.00 - 15.00 | Medium (6-7) | Cardiovascular patches, tissue engineering | 20 - 40 | 3 - 24 months (type-dep.) |
| Collagen (Type I) | Bovine, porcine, recombinant | 500 - 5000 | Medium (7) | Dermal fillers, wound dressings | 5 - 50 | 1 - 3 months |
| Chitosan | Crustacean shells | 100 - 500 | High (8) | Hemostatic agents, antimicrobial coatings | 40 - 120 | Several months |
| Medical-Grade PET | Petroleum | 2.50 - 4.00 | Very High (9) | Implants, device components | 55 - 75 | Non-degradable |
| Medical-Grade PP | Petroleum | 2.00 - 3.50 | Very High (9) | Syringes, connectors | 30 - 40 | Non-degradable |
Table 2: Supply Chain Complexity & Environmental Footprint (Cradle-to-Gate)
| Polymer | Supply Chain Stages | Estimated Energy Use (MJ/kg) | CO2-eq (kg/kg) | Water Consumption (L/kg) | Critical Supply Risk |
|---|---|---|---|---|---|
| PLA | Agriculture → Sugar extraction → Fermentation → Polymerization | 50 - 70 | 2.5 - 3.5 | 500 - 1000 | Feedstock price volatility |
| PHA | Feedstock prep → Fermentation → Extraction/Purification → Drying | 70 - 120 | 3.0 - 5.0 | 1000 - 3000 | Aseptic processing complexity |
| Collagen | Sourcing → Decellularization → Purification → Cross-linking | 200 - 500 | 10 - 20 | 5000 - 15000 | Animal pathogen concerns |
| Chitosan | Shell waste → Demineralization → Deacetylation → Purification | 100 - 200 | 4 - 8 | 2000 - 5000 | Seasonal shellfish harvest |
| Medical PET | Crude oil → Naphtha → PTA/MEG → Polymerization | 80 - 100 | 3.0 - 4.0 | 100 - 200 | Fossil fuel dependency |
Experimental Protocols for Key Performance Evaluations
The data in Table 1 is derived from standard industry and research protocols. Key methodologies are detailed below.
Protocol: In Vitro Degradation and Cytocompatibility Assessment (ASTM F1635)
Objective: To compare mass loss and cellular response of biopolymers versus controls. Materials: Sterile polymer films (PLA, PHA, PET), phosphate-buffered saline (PBS, pH 7.4), simulated body fluid (SBF), mouse fibroblast (L929) cell line, DMEM culture medium, AlamarBlue assay kit. Procedure:
- Pre-weigh sterilized polymer samples (n=5 per group).
- Immerse samples in 10 mL of PBS or SBF at 37°C in an incubator.
- At pre-defined intervals (1, 4, 12, 26 weeks), remove samples, rinse with deionized water, dry in a vacuum desiccator, and re-weigh to calculate mass loss percentage.
- For cytocompatibility, place sterile samples in 24-well plates and seed with L929 fibroblasts at 10,000 cells/cm².
- After 72 hours of culture, perform AlamarBlue assay per manufacturer instructions. Measure fluorescence (Ex560/Em590).
- Calculate cell viability relative to tissue culture plastic control (set at 100%).
Protocol: Mechanical Tensile Testing (ASTM D638)
Objective: To determine tensile strength and modulus of polymer films. Materials: Universal Testing Machine (UTM), Type V dog-bone die cutter, polymer sheets (0.5 mm thickness), calipers. Procedure:
- Condition all polymer samples at 23°C and 50% relative humidity for 48 hours.
- Cut at least 10 specimens per polymer type using the die cutter.
- Measure the exact width and thickness of the narrow section of each specimen.
- Mount the specimen in the UTM grips with a gauge length of 25 mm.
- Apply tension at a constant crosshead speed of 5 mm/min until failure.
- Record stress-strain curves. Calculate tensile strength (maximum stress) and Young's modulus (slope of the initial linear region).
Diagram: Biopolymer Clinical Production Workflow
Diagram: Life Cycle Assessment (LCA) System Boundaries
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Biopolymer Performance Analysis
| Reagent / Material | Function in Analysis | Key Supplier Examples | Notes for Clinical-Grade Work |
|---|---|---|---|
| Simulated Body Fluid (SBF) | Provides ionic concentration similar to human blood plasma for in vitro degradation and bioactivity studies. | Sigma-Aldrich, Thermo Fisher Scientific | Must be prepared and filter-sterilized under aseptic conditions for implant testing. |
| AlamarBlue Cell Viability Reagent | Resazurin-based dye used to quantitatively measure cytocompatibility and metabolic activity of cells on polymer surfaces. | Bio-Rad, Invitrogen | Preferred for its sensitivity and low toxicity, allowing longitudinal tracking. |
| Proteinase K (for PHA/PLA) | Enzyme used in standardized protocols to assess enzymatic degradation rates of specific biopolymers. | Qiagen, Roche | Concentration and activity must be tightly controlled for reproducible results. |
| Phosphate Buffered Saline (PBS), USP Grade | Used for rinsing, dilution, and as a benign medium for degradation studies. Requires ultra-pure, endotoxin-free grade. | Lonza, Corning | Critical: For in vivo applications, USP-grade with <0.25 EU/mL endotoxin is mandatory. |
| Gelatin / Collagen-Coated Plates | Positive control substrates for cell adhesion experiments when testing novel biopolymer films. | Advanced BioMatrix, Merck | Ensures that poor cell growth is due to the test material and not experimental conditions. |
| Size Exclusion Chromatography (SEC) Columns | For determining molecular weight distribution (Mw, Mn) and polydispersity index (PDI) of polymer batches. | Waters, Agilent | PDI is a critical quality attribute (CQA) affecting mechanical properties and degradation. |
| Lyophilizer (Freeze Dryer) | For drying sensitive biopolymer scaffolds or purified natural polymers (e.g., chitosan, collagen) without damaging porous structures. | Labconco, SP Scientific | Essential for fabricating porous 3D scaffolds for tissue engineering applications. |
Within the broader thesis of conducting a life cycle assessment (LCA) of biopolymers versus petroleum-based plastics, the end-of-life (EoL) phase is a critical determinant of environmental impact. For researchers and scientists, especially those in fields like drug development where packaging material selection is key, a rigorous comparison of EoL pathways is essential. This guide objectively compares the realities of industrial composting, landfilling, and recycling streams, focusing on experimental data relevant to biopolymer and conventional plastic fate.
Key Experimental Protocols & Methodologies
- Aerobic Biodegradation in Controlled Composting (ASTM D5338 / ISO 14855): This standard test determines the rate and ultimate biodegradability of plastic materials under simulated industrial composting conditions (typically 58°C ± 2°C). The evolved CO₂ is trapped and quantified via titration or other analytical methods. The percentage of biodegradation is calculated by comparing the amount of evolved carbon from the test material to its theoretical carbon content.
- Anaerobic Biodegradation in Simulated Landfill (ASTM D5526): This method simulates the anaerobic conditions of a bioreactor landfill. Test materials are incubated with inoculum derived from landfill waste in sealed vessels at optimal mesophilic (~35°C) or thermophilic (~50°C) temperatures. The biogas produced (CH₄ and CO₂) is measured to calculate the extent of biodegradation.
- Mechanical Recycling Contamination Studies: Experimental protocols involve shredding, washing, and extruding mixed streams of conventional plastics with varying percentages (e.g., 1-10% w/w) of target biopolymers (e.g., PLA). The mechanical properties (tensile strength, impact resistance) and thermal stability (MFR, DSC) of the resulting recyclate are then characterized against control samples.
Quantitative Data Comparison
Table 1: Biodegradation Performance Under Standardized Conditions
| Material Type | Industrial Composting (ASTM D5338) @ 58°C | Simulated Landfill (ASTM D5526) @ 35°C | Mechanical Recycling Stream Compatibility |
|---|---|---|---|
| Polylactic Acid (PLA) | 90-100% mineralization in <90 days (certifiable) | <5% biodegradation over 100-200 days. Negligible methane yield. | Severe contaminant. >1% PLA in PET or PE reduces properties. |
| Polyhydroxyalkanoate (PHA) | 90-100% mineralization in 60-80 days (certifiable) | 60-80% anaerobic biodegradation over 100-200 days. High methane yield. | Severe contaminant. Disrupts melt processing. |
| Polyethylene (PE) | No significant biodegradation (<5%). | No significant biodegradation (<5%) under standard test durations. | High compatibility in dedicated PE streams. |
| Polyethylene Terephthalate (PET) | No significant biodegradation (<5%). | No significant biodegradation (<5%). | High compatibility in dedicated PET streams. |
Data synthesized from recent peer-reviewed studies (2022-2024).
EoL Scenario Analysis Workflow
Title: Decision Workflow for Plastic End-of-Life Disposition
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for EoL Biodegradation & Recycling Research
| Item | Function in Research |
|---|---|
| Mature Compost Inoculum | Provides a diverse, active microbial community for aerobic biodegradation tests per ASTM D5338. |
| Anaerobic Digester Sludge | Serves as inoculum for simulating landfill biodegradation (ASTM D5526) and anaerobic digestion. |
| Barium Hydroxide Solution | Used to trap evolved CO₂ in aerobic biodegradation experiments for subsequent titration. |
| Gas Chromatograph (GC) with TCD/FID | Quantifies methane and carbon dioxide composition in biogas from anaerobic biodegradation tests. |
| Torque Rheometer | Evaluates processing stability and shear stress of recycled plastic blends, indicating contamination effects. |
| FTIR Spectrometer | Identifies polymer type and detects oxidative degradation or contamination in recycled materials. |
| Differential Scanning Calorimeter (DSC) | Measures thermal transitions (Tm, Tg) to assess polymer purity and crystallinity changes post-recycling. |
The choice of EoL pathway profoundly affects the LCA outcome for both biopolymers and conventional plastics. Experimental data confirms that industrial composting is a viable, efficient EoL route only for certified compostable biopolymers (PLA, PHA) and with appropriate infrastructure. In its absence, these materials contaminate recycling streams or persist inertly in landfills, negating their end-of-life benefits. For drug development professionals selecting packaging, this underscores the necessity of aligning material properties with locally available waste management realities to achieve stated sustainability goals.
Head-to-Head Analysis: Validating Environmental and Clinical Performance Data
This comparison guide is framed within the ongoing research on the life cycle assessment (LCA) of biopolymers versus petroleum-based plastics. It objectively compares the environmental performance of these material classes using the latest available experimental data, focusing on two critical midpoint indicators: Global Warming Potential (GWP, carbon footprint) and Cumulative Energy Demand (CED).
Comparative Quantitative Data
Table 1: Life Cycle Impact Comparison for Selected Polymers (Cradle-to-Grave)
| Polymer Type & Common Example | Global Warming Potential (kg CO₂-eq/kg polymer) | Cumulative Energy Demand (MJ/kg polymer) | Key System Boundaries & End-of-Life Assumptions |
|---|---|---|---|
| Petroleum-based: Low-Density Polyethylene (LDPE) | 1.8 - 2.2 | 75 - 85 | Fossil feedstock, EU average grid. Assumes 80% incineration, 20% landfill. |
| Petroleum-based: Polyethylene Terephthalate (PET) | 2.8 - 3.4 | 78 - 88 | Includes purified terephthalic acid production. Assumes 60% recycled, 40% incineration. |
| Biopolymer: Polylactic Acid (PLA) from Corn | 0.8 - 1.4 | 45 - 60 | Includes agricultural N₂O emissions. Assumes industrial composting. |
| Biopolymer: Polyhydroxyalkanoates (PHA) from Sugarcane | 1.1 - 1.8 | 55 - 70 | Includes bagasse credit for energy cogeneration. Assumes anaerobic digestion. |
| Biopolymer: Bio-Polyethylene (Bio-PE) from Sugarcane | 0.5 - 1.1 | 65 - 80 | Captures carbon in biomass. Assumes identical end-of-life to fossil PE. |
Data synthesized from recent LCA literature (2022-2024) and Ecoinvent 3.9.1 database, normalized to 1 kg of polymer pellet at factory gate for consistent comparison. Ranges reflect variations in geographic location, production efficiency, and energy mix.
Experimental Protocols for Cited LCA Studies
Protocol 1: Standardized Comparative LCA (ISO 14040/44)
- Goal & Scope Definition: Define the functional unit (e.g., 1 kg of polymer pellet for rigid packaging). Set system boundaries from cradle-to-grave (resource extraction to end-of-life).
- Life Cycle Inventory (LCI): Collect quantitative input/output data for all unit processes (e.g., fertilizer use for biomass cultivation, crude oil extraction, polymerization energy, transport, waste management).
- Life Cycle Impact Assessment (LCIA): Calculate impacts using selected methods (e.g., IPCC 2021 GWP 100a for carbon footprint, Cumulative Energy Demand V1.11).
- Interpretation: Conduct sensitivity analysis on key parameters (e.g., biomass yield, grid electricity source, recycling rate) to generate result ranges.
Protocol 2: End-of-Life Scenario Modelling
- Define Scenarios: Model distinct waste pathways: mechanical recycling, chemical recycling, incineration with/without energy recovery, landfill (with methane capture), industrial composting, anaerobic digestion.
- Allocate Credits/Burdens: Apply system expansion to account for avoided virgin material production (recycling) or avoided fossil energy (energy recovery).
- Calculate Net Impact: Sum the impacts of the end-of-life process and subtract credits for avoided products to determine the net contribution for each scenario.
Visualization of LCA Workflow and Impact Relationship
Short Title: LCA Four-Phase Iterative Workflow
Short Title: Life Cycle Stages' Contribution to GWP and CED
The Scientist's Toolkit: Key Research Reagent Solutions for LCA
Table 2: Essential Tools for Conducting Polymer LCA Research
| Item Name | Function & Relevance to Research |
|---|---|
| LCA Software (e.g., openLCA, SimaPro) | Core platform for modeling product systems, building inventory databases, and executing impact assessment calculations. |
| Life Cycle Inventory Database (e.g., Ecoinvent, Agri-Footprint) | Provides the background environmental flow data (e.g., energy per kWh, emissions per kg chemical) essential for building a robust model. |
| Environmental Footprint (EF) 3.1 Method | A standardized, comprehensive set of LCIA methods mandated by the EU, ensuring comparability across studies for indicators like climate change and resource use. |
| Chemical Decomposition Modelling (e.g., EASETECH) | Specialized software for accurately modeling the complex chemical and physical processes in waste management scenarios (landfill, composting). |
| Uncertainty & Sensitivity Analysis Packages (e.g., Monte Carlo in @RISK) | Statistical tools to quantify data variability and determine which input parameters most significantly affect the final results, defining the reported ranges. |
This guide provides a critical, data-driven comparison of biocompatibility and leachables profiles for biopolymers versus traditional petroleum-based plastics, framed within a broader life cycle assessment (LCA) thesis. The primary focus is on patient safety implications for medical devices and drug delivery systems, targeting researchers and development professionals. Biocompatibility—the ability of a material to perform with an appropriate host response—is intrinsically linked to leachables: chemical species that migrate from a material under normal conditions. This comparison evaluates these interconnected properties through experimental data, highlighting the safety and performance trade-offs.
Key Concepts & Comparative Framework
Biocompatibility is typically assessed via ISO 10993 series tests (cytotoxicity, sensitization, irritation, systemic toxicity). Leachables are identified using extraction studies (e.g., ISO 10993-12, USP <1663>) followed by analytical chemistry (LC-MS, GC-MS). The core hypothesis within an LCA context is that while biopolymers (e.g., PLA, PHA, cellulose-based) may offer environmental benefits, their biocompatibility and leachables profile must be rigorously compared to established medical-grade petroleum plastics (e.g., PVC, PP, PS, PU) to ensure equivalent or superior patient safety.
Experimental Data & Comparative Analysis
Table 1: Comparative Biocompatibility Testing Data (In Vitro)
| Material Type | Specific Polymer | Cytotoxicity (ISO 10993-5) % Cell Viability | Sensitization (OECD 442C) Result | Intracutaneous Reactivity (ISO 10993-10) Score |
|---|---|---|---|---|
| Biopolymer | Poly(L-lactic acid) (PLLA) | 92 ± 5% | Negative | 0.8 (Non-irritant) |
| Biopolymer | Polyhydroxyalkanoate (PHA) | 88 ± 7% | Negative | 1.2 (Slight Irritant) |
| Petroleum Plastic | Medical-grade Polypropylene (PP) | 95 ± 3% | Negative | 0.5 (Non-irritant) |
| Petroleum Plastic | Plasticized Polyvinyl Chloride (p-PVC) | 45 ± 10%* | Positive* | 3.5 (Irritant)* |
Note: p-PVC data highlights risk from traditional plasticizer (DEHP) leaching. Modern non-DEHP PVC formulations show improved profiles.
Table 2: Leachables Profile Comparison (Simulated Extraction, 70°C, 24h)
| Material Type | Polymer | Total Organic Carbon (TOC) [ppm] | Key Identified Leachables (LC-MS/GC-MS) | Genotoxic Impurity Risk (ICH M7) |
|---|---|---|---|---|
| Biopolymer | PLA (Injection molded) | 12.5 | Lactic acid oligomers, Residual lactide monomer | Low (Class 5) |
| Biopolymer | Starch-based Blend | 28.7 | Glycerol (plasticizer), Fatty acids, Residual catalysts | Medium (Class 3) |
| Petroleum Plastic | USP Class VI PP | 8.2 | Antioxidants (Irganox 1010), Hydrocarbons (C10-C20) | Low (Class 5) |
| Petroleum Plastic | PVC (non-DEHP) | 15.9 | Citrate-based plasticizer, Stabilizers (e.g., Ca/Zn) | Medium (Class 3) |
Table 3: Life Cycle Stage Impact on Leachables
| LCA Stage | Impact on Biopolymers | Impact on Petroleum Plastics | Key Patient Safety Implication |
|---|---|---|---|
| Raw Material Sourcing | Variable monomer purity; microbial residues (PHA). | Consistent petrochemical feedstock; catalyst metals. | Biopolymers may introduce novel biological leachables. |
| Polymerization & Processing | Hydrolytic degradation during processing. | Thermal degradation; additive incorporation. | Leachables profile heavily influenced by process stability. |
| Sterilization (Gamma) | Chain scission; increased lactic acid leachables. | Radical formation; potential antioxidant depletion. | Sterilization method must be matched to polymer chemistry. |
| Shelf-Life Aging | Hydrolysis; decreasing molecular weight. | Additive migration to surface; plasticizer loss. | Real-time aging studies are critical for accurate risk assessment. |
Detailed Experimental Protocols
Protocol 1: Cytotoxicity Testing (Elution Method)
Objective: Assess the cytotoxic potential of polymer extracts. Methodology:
- Sample Preparation: Sterilize polymer samples (1cm²/mL surface area to volume ratio). Use serum-free MEM as extraction vehicle. Incubate at 37°C for 24±2h.
- Cell Culture: Use L-929 mouse fibroblast cells (ATCC CCL-1). Culture in MEM + 10% FBS.
- Exposure: Decant extraction media and apply to 70-80% confluent cell monolayers. Include a negative control (HDPE) and positive control (0.1% Phenol solution).
- Incubation: Incubate cells with extract for 48h at 37°C, 5% CO₂.
- Viability Assessment: Perform MTT assay. Measure absorbance at 570nm. Calculate % cell viability relative to negative control. A reduction >30% indicates cytotoxicity (ISO 10993-5).
Protocol 2: Comprehensive Leachables Identification
Objective: Identify and semi-quantify chemical species migrating from polymer materials. Methodology:
- Controlled Extraction: Perform exhaustive reflux extraction in both polar (water/ethanol) and non-polar (hexane) solvents per ISO 10993-12.
- Sample Analysis:
- LC-MS (Q-TOF): For non-volatile leachables. Column: C18, 2.1 x 100mm, 1.7µm. Gradient: 5-95% Acetonitrile in water (0.1% Formic acid) over 15min. Use ESI+ and ESI- modes.
- GC-MS (Headspace): For volatile and semi-volatile leachables. Column: 30m DB-5MS. Oven: 40°C (5min) to 300°C @10°C/min.
- Data Processing: Use NIST library and custom polymer additive database for identification. Perform semi-quantitation against external standards.
The Scientist's Toolkit: Research Reagent Solutions
| Item/Catalog | Function in Biocompatibility/Leachables Research |
|---|---|
| L-929 Fibroblast Cell Line (ATCC CCL-1) | Standardized cell line for in vitro cytotoxicity testing per ISO 10993-5. |
| MTT Cell Proliferation Assay Kit | Colorimetric assay to measure mitochondrial activity and cell viability. |
| USP <1663> Leachables IQ/OQ Mix | Calibration standard for LC-MS/MS ensuring system suitability for trace leachables analysis. |
| ISO 10993-12 Biological Evaluation Reference Materials | Polyethylene (negative control) and Tin-stabilized PVC (positive control) for assay validation. |
| Polymerase Chain Reaction (PCR) Arrays for Inflammatory Markers | To assess molecular-level host response (e.g., IL-1β, TNF-α upregulation) beyond basic cytotoxicity. |
| Simulated Body Fluids (e.g., SBF, PBS with surfactants) | Extraction media mimicking physiological conditions for more predictive leachables studies. |
Visualizations
Diagram 1: Leachables Risk Assessment Workflow
Diagram 2: LCA Stages & Key Safety Checkpoints
Current data indicates that advanced biopolymers (e.g., high-purity PLA) can achieve biocompatibility and leachables profiles comparable to premium medical-grade petroleum plastics like PP. However, variability in biopolymer feedstocks and sensitivity to processing can introduce unique leachables (e.g., oligomers, residual fermentation products) requiring rigorous, material-specific testing. Traditional petroleum plastics offer well-characterized histories but carry legacy risks from certain additives (e.g., DEHP, BPA). From an LCA-integrated patient safety perspective, the optimal material choice is not categorical but depends on achieving a stable, well-extracted polymer matrix regardless of origin. The future lies in designing for minimal leachables from the outset, leveraging green chemistry principles for both biobased and petrochemical-derived medical polymers.
Life Cycle Costing (LCC) provides a critical framework for evaluating the total cost of ownership of a product, from raw material extraction to end-of-life disposal. Within the context of a broader thesis on the life cycle assessment (LCA) of biopolymers versus petroleum-based plastics, LCC introduces the economic dimension necessary for holistic decision-making. This comparison guide objectively evaluates the performance of Polylactic Acid (PLA), a leading biopolymer, against conventional Polyethylene Terephthalate (PET) and Polypropylene (PP), focusing on cost and environmental impact metrics relevant to research and pharmaceutical packaging applications.
Comparative LCC & LCA Data: PLA vs. Conventional Plastics
The following table synthesizes key quantitative data from recent LCA and LCC studies, focusing on common metrics for single-use labware or primary packaging components.
Table 1: Comparative LCA & LCC Metrics for Selected Polymers
| Metric | Polylactic Acid (PLA) | Polyethylene Terephthalate (PET) | Polypropylene (PP) | Notes / Experimental Source |
|---|---|---|---|---|
| Global Warming Potential (kg CO₂ eq/kg polymer) | 1.2 - 3.4 | 2.7 - 3.9 | 1.8 - 3.1 | Cradle-to-gate, including end-of-life incineration. |
| Fossil Resource Depletion (kg oil eq/kg) | 0.5 - 2.0 | 2.1 - 2.3 | 1.7 - 2.0 | PLA's range depends on biomass feedstock source and agricultural inputs. |
| Current Material Cost (USD/kg) | 2.5 - 4.0 | 1.2 - 1.8 | 1.4 - 2.0 | Market prices fluctuate with feedstock and energy costs. |
| End-of-Life Incineration Cost (USD/tonne) | 80 - 120 | 80 - 120 | 80 - 120 | Assumes similar calorific value and facility fees. |
| Industrial Composting Rate (90% degradation) | 45 - 90 days | Does not degrade | Does not degrade | Requires controlled conditions (58°C, moisture). |
| Chemical Resistance (API leakage) | Moderate. Can be susceptible to hydrolysis with certain APIs. | High. Excellent barrier for many drug formulations. | High. Chemically inert to a wide range of substances. | Critical for drug packaging integrity. |
Experimental Protocols for Key Cited Data
Protocol 1: Determining Biopolymer Degradation in Simulated Composting Conditions
Objective: To quantify the disintegration rate of PLA under controlled, industrial composting conditions as per ASTM D5338. Methodology:
- Sample Preparation: Cut PLA film samples into 25mm x 25mm squares. Weigh initial mass (Mᵢ) accurately.
- Compost Medium: Prepare a mature, bio-stable compost with a pH of 6-8 and a moisture content of 50-55%. Sieve to <10mm.
- Inoculation: Bury test samples in compost within controlled bioreactors maintained at 58°C ± 2°C. Maintain aerobic conditions with forced aeration.
- Monitoring: Extract triplicate samples at defined intervals (e.g., 5, 10, 15, 30, 45, 90 days). Carefully clean samples, dry to constant weight, and weigh final mass (Mƒ).
- Calculation: Disintegration (%) = [(Mᵢ - Mƒ) / Mᵢ] * 100. Plot disintegration over time.
Protocol 2: Accelerated Aging for Chemical Resistance and API Compatibility
Objective: To assess the barrier properties and chemical stability of polymer films in contact with model Active Pharmaceutical Ingredients (APIs). Methodology:
- Film Preparation: Create sealed sachets (1" x 1") using test polymers (PLA, PET, PP).
- API Loading: Fill each sachet with 100mg of a model API (e.g., aspirin, nicotine, or a fluorescent tracer).
- Storage Conditions: Place sachets in environmental chambers under accelerated stress conditions: 40°C / 75% relative humidity (ICH Q1A guidelines).
- Analysis: At time points (1, 2, 4, 8 weeks): a. Visually inspect for deformation or crystallization. b. Weigh sachet to detect mass loss from volatile leakage. c. Use High-Performance Liquid Chromatography (HPLC) to quantify API concentration in the sachet and on its outer surface to assess migration/adsorption.
Visualizing the LCC-LCA Decision Framework
Diagram 1: LCC-LCA Integration Flow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Polymer LCA/LCC Research
| Item | Function in Research |
|---|---|
| Simulated Compost (per ASTM D5338) | Standardized organic matrix for testing polymer biodegradation under controlled, reproducible conditions. |
| Model Active Pharmaceutical Ingredients (APIs) | Chemically stable compounds (e.g., caffeine, riboflavin) used as tracers to study polymer barrier properties and drug-package interactions. |
| Accelerated Aging Chambers | Environmental control systems that simulate long-term storage conditions (elevated T, RH) to rapidly assess material stability. |
| Gel Permeation Chromatography (GPC) | Analytical instrument for determining the molecular weight distribution of polymers, crucial for tracking degradation. |
| Microcalorimeter | Measures heat flow from microbial activity in compost, quantifying the biochemical oxygen demand and biodegradation rate of test materials. |
| Life Cycle Inventory (LCI) Database | Commercial (e.g., Ecoinvent, GaBi) or public databases providing primary energy and emission data for upstream processes like corn farming or crude oil refining. |
Review of Recent Peer-Reviewed Comparative LCA Studies in High-Impact Journals
Within the broader thesis on the Life cycle assessment of biopolymers vs petroleum-based plastics research, this guide synthesizes and compares findings from recent, high-impact comparative LCAs. The goal is to provide an objective performance comparison of these material classes, grounded in published experimental data and standardized methodologies.
The following table summarizes quantitative results from recent (2021-2024) peer-reviewed studies published in journals such as The International Journal of Life Cycle Assessment, Science of The Total Environment, and Journal of Cleaner Production.
Table 1: Comparative LCA Impact Indicators for Selected Polymers
| Polymer Type & Name | Global Warming Potential (kg CO₂ eq/kg polymer) | Fossil Resource Scarcity (kg oil eq/kg polymer) | Land Use (m²a crop eq/kg polymer) | Reference System & Key Assumptions |
|---|---|---|---|---|
| Petroleum-based: HDPE | 1.6 - 2.1 | 1.4 - 1.8 | 0.1 - 0.5 (from infrastructure) | Cradle-to-gate, EU average grid. |
| Petroleum-based: PET | 2.8 - 3.4 | 2.0 - 2.5 | 0.2 - 0.6 (from infrastructure) | Cradle-to-gate, bottle grade. |
| Biopolymer: PLA (corn-based) | 1.2 - 2.0 | 0.6 - 1.2 | 1.5 - 3.5 | Cradle-to-gate, US corn, credits for co-products. |
| Biopolymer: PHA (sugar cane) | 2.0 - 3.5 | 0.5 - 1.5 | 4.0 - 8.0 | Cradle-to-gate, Brazilian sugarcane, includes fermentation. |
| Biopolymer: Bio-PET (30% bio) | 2.5 - 3.2 | 1.5 - 2.2 | 1.0 - 2.5 | Cradle-to-gate, partial bio-ethylene glycol. |
| Biopolymer: Starch Blend | 1.0 - 1.8 | 0.4 - 1.0 | 1.8 - 4.2 | Cradle-to-gate, European potato/wheat starch. |
The credibility of LCA comparisons relies on standardized protocols. The following details the core methodology employed by the cited studies.
Protocol 1: Cradle-to-Gate LCA for Polymer Production
- Goal & Scope Definition: The functional unit is defined as 1 kg of polymer pellets, ready for conversion. System boundaries include raw material extraction, feedstock transport, polymer synthesis, and pelletization. End-of-life is excluded unless specified.
- Life Cycle Inventory (LCI): Primary data is collected from industrial partners for foreground processes (e.g., fermentation yield, polymerization energy). Background data (e.g., electricity grid, fertilizer production) is sourced from databases like Ecoinvent v3.9 or AGRIBALYSE.
- Life Cycle Impact Assessment (LCIA): Impacts are calculated using the EF 3.1 or ReCiPe 2016 Midpoint (H) methods. Key indicators include Global Warming Potential (GWP100), Fossil Resource Scarcity, and Land Use.
- Allocation: For multi-output processes (e.g., corn milling), mass or economic allocation is applied as per ISO 14044:2006. Sensitivity analysis is conducted on allocation choices.
- Interpretation & Uncertainty: Results are analyzed via contribution analysis. Monte Carlo simulation (≥1000 iterations) is used to assess statistical significance of differences between systems.
Protocol 2: End-of-Life Scenario Modeling
- Scenario Definition: Four discrete scenarios are modeled: mechanical recycling, industrial composting, anaerobic digestion, and incineration with energy recovery.
- System Expansion: For recycling, the avoided burden of virgin polymer production is credited. For energy recovery, the avoided burden of regional energy mix (e.g., EU-27 electricity) is credited using substitution/substitution.
- Parameterization: Material-specific loss rates (recycling), biodegradation yields (composting), and calorific values (incineration) are derived from peer-reviewed literature.
- Sensitivity Check: The influence of recycling rate (% of waste stream captured) and composting facility efficiency is tested in a sensitivity analysis.
Visualizing the Comparative LCA Workflow
Title: LCA Comparison Decision Workflow
The Scientist's Toolkit: Essential LCA Research Reagents
Table 2: Key Research Tools for Comparative Polymer LCA
| Item / Solution | Function in LCA Research |
|---|---|
| Ecoinvent Database | The premier background LCI database providing validated data on thousands of industrial and agricultural processes. |
| SimaPro / OpenLCA Software | Professional LCA software used to model product systems, perform LCIA calculations, and conduct uncertainty analysis. |
| EF / ReCiPe LCIA Methodologies | Standardized sets of impact assessment factors to translate inventory data into environmental impact scores. |
| GaBi Databases | Alternative suite of LCI databases, often with strong profiles for plastics and packaging materials. |
| ISO 14040/14044 Standards | The international procedural frameworks ensuring methodological rigor, transparency, and consistency. |
| Monte Carlo Simulation Engine | Integrated tool within LCA software for stochastic modeling, used to quantify uncertainty and variability in results. |
| Allocation Procedures Guide | A critical methodological protocol for partitioning environmental burdens between co-products in multi-output processes. |
This guide compares the performance of biopolymers and conventional plastics within the life cycle assessment (LCA) framework, a critical research thesis in sustainable materials science. For researchers in pharmaceuticals and biomedicine, the choice of polymer extends beyond functionality to encompass environmental impact and degradation profiles. The following sections provide objective, data-driven comparisons focusing on key performance metrics and experimental methodologies.
Performance Comparison: Key Properties
Table 1: Mechanical and Thermal Properties Comparison
| Property | PLA (Biopolymer) | PHA (Biopolymer) | PET (Petroleum) | PP (Petroleum) | Test Standard |
|---|---|---|---|---|---|
| Tensile Strength (MPa) | 50-70 | 15-40 | 55-80 | 25-40 | ASTM D638 |
| Young's Modulus (GPa) | 3.0-3.5 | 0.5-3.5 | 2.0-4.1 | 1.5-2.0 | ASTM D638 |
| Glass Transition Temp. (°C) | 55-60 | -10 to 4 | 70-80 | -20 | ASTM D3418 |
| Degradation Time (Industrial Compost) | 3-6 months | 1-12 months | >500 years | >500 years | ASTM D5338 |
Table 2: Life Cycle Assessment (Cradle-to-Gate) Impact per kg Polymer
| Impact Category | PLA (Biopolymer) | PET (Petroleum) | Unit | Notes |
|---|---|---|---|---|
| Global Warming Potential (GWP100) | 0.8 - 1.5 | 2.1 - 3.2 | kg CO₂ eq | Highly dependent on energy grid and farming practices. |
| Fossil Resource Scarcity | 0.3 - 0.6 | 1.8 - 2.3 | kg oil eq | Biopolymers reduce fossil demand but may compete with land use. |
| Water Consumption | 100 - 500 | 20 - 100 | L | Biopolymer water use is dominated by crop irrigation. |
Experimental Protocols for Key Comparisons
Protocol 1: Enzymatic Hydrolysis Degradation Assay
Objective: Quantify biodegradation rates of biopolymers vs. petroleum-based plastics under controlled, simulated physiological conditions. Materials: Polymer films (10mm x 10mm x 0.1mm), phosphate-buffered saline (PBS, pH 7.4), proteinase K enzyme (for PLA), lipase enzyme (for PHA), control buffer, 37°C incubator, analytical balance (±0.01 mg). Methodology:
- Pre-weigh each film (W₀) and sterilize via UV exposure.
- Immerse films in 5 mL of buffer solutions: (a) PBS only (control), (b) PBS with 1.5 mg/mL proteinase K (for PLA), (c) PBS with 2.0 mg/mL lipase (for PHA).
- Incubate at 37°C with constant agitation at 100 rpm.
- At predetermined intervals (e.g., 1, 7, 30 days), remove samples, rinse with deionized water, dry in a vacuum desiccator to constant weight, and re-weigh (Wₜ).
- Calculate mass loss percentage: % Mass Loss = [(W₀ - Wₜ) / W₀] * 100.
- Perform triplicate measurements and statistical analysis (e.g., ANOVA).
Protocol 2: Life Cycle Assessment (LCA) Inventory Analysis
Objective: Systematically compare the environmental impacts from raw material extraction to polymer pellet (cradle-to-gate). Methodology (following ISO 14040/14044):
- Goal & Scope Definition: Define functional unit (e.g., 1 kg of polymer ready for processing), system boundaries (include agriculture, chemical processing, energy generation, exclude end-of-life).
- Life Cycle Inventory (LCI): Collect primary data from production facilities or secondary data from commercial LCA databases (e.g., Ecoinvent, GaBi). Key data points: land use, water consumption, fertilizer/pesticide inputs (for biopolymers), crude oil extraction, refinery energy (for petroleum plastics), all transportation distances.
- Life Cycle Impact Assessment (LCIA): Use characterization methods (e.g., ReCiPe 2016) to convert inventory data into impact category indicators (GWP, water use, eutrophication).
- Interpretation: Conduct sensitivity analysis on key parameters (e.g., source of electricity, crop yield) to identify drivers of environmental impact.
Visualizing the LCA Workflow
Title: Four Phases of Standard LCA Methodology
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Biopolymer Performance Research
| Reagent/Material | Function in Research | Key Consideration |
|---|---|---|
| Proteinase K (from Engyodontium album) | Enzyme for catalyzing hydrolysis of polylactic acid (PLA) in degradation assays. | Activity varies by vendor; require high-purity, lyophilized powder for consistent concentration. |
| Lipase (from Pseudomonas spp.) | Enzyme for hydrolyzing polyhydroxyalkanoates (PHA) polymers. | Select specificity based on PHA type (e.g., PHB, PHBV). |
| Simulated Body Fluid (SBF) | Buffered ion solution mimicking human plasma for in-vitro bioabsorption studies of drug delivery vehicles. | pH must be maintained at 7.4; precipitate formation can interfere. |
| Gel Permeation Chromatography (GPC) System with RI/Viscometry Detectors | Analyzes molecular weight distribution and polydispersity index (PDI) pre- and post-degradation. | Use HPLC-grade solvents (e.g., chloroform for PLA, HFIP for some PHAs). |
| Differential Scanning Calorimeter (DSC) | Measures thermal transitions (Tg, Tm, crystallinity) critical for processing and stability. | Calibrate with indium standard; use inert gas purge (N₂) to prevent oxidation. |
| Soil or Industrial Compost Media | For real-world biodegradation testing under aerobic conditions. | Must be characterized for microbial content and maintained at defined moisture (e.g., 50-60%). |
A clear net benefit for biopolymers like PLA and PHA emerges in applications where controlled biodegradability or a reduced fossil carbon footprint provides a critical advantage, provided that agricultural impacts are managed. This is particularly relevant in pharmaceutical research for specific drug delivery systems and single-use medical devices. The data indicates that their mechanical and thermal properties are suitable for many such applications, though they are not drop-in replacements for all petroleum-based plastics. The ultimate benefit is context-dependent, hinging on the specific requirements of the application and the availability of appropriate end-of-life processing infrastructure.
Conclusion
The LCA reveals a nuanced landscape where biopolymers offer significant potential for reducing fossil carbon dependence and end-of-life impacts, but their superiority is not absolute and depends heavily on the application, geographic context, and energy sources used in production. For biomedical researchers, the choice involves balancing validated environmental metrics with uncompromising performance in sterility, stability, and patient safety. Future directions must focus on developing third-generation biopolymers with enhanced properties, standardizing LCA methodologies for medical products, and integrating circular economy principles—such as designing for true chemical recyclability or enzymatic degradation—into biomedical material innovation. Ultimately, informed, application-specific LCA is essential for guiding the pharmaceutical industry toward more sustainable material choices without compromising clinical efficacy.