Ultimate Guide to MALDI-TOF MS for Polymer Molecular Weight Analysis: From Fundamentals to Advanced Applications
This comprehensive guide demystifies Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) for polymer characterization.
Ultimate Guide to MALDI-TOF MS for Polymer Molecular Weight Analysis: From Fundamentals to Advanced Applications
Abstract
This comprehensive guide demystifies Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) for polymer characterization. Tailored for researchers, scientists, and drug development professionals, it covers foundational principles, step-by-step methodologies for synthetic and biopolymers, and critical troubleshooting for complex samples like polyesters and polyethers. We explore advanced optimization techniques, compare MALDI-TOF MS to SEC and ESI-MS for accuracy and limitations, and validate its role in determining absolute molecular weight (Mn, Mw), dispersity (Đ), and end-group analysis. This article equips readers to implement robust, high-resolution polymer analysis for materials science and pharmaceutical development.
What is MALDI-TOF MS for Polymers? Understanding Core Principles and Key Metrics
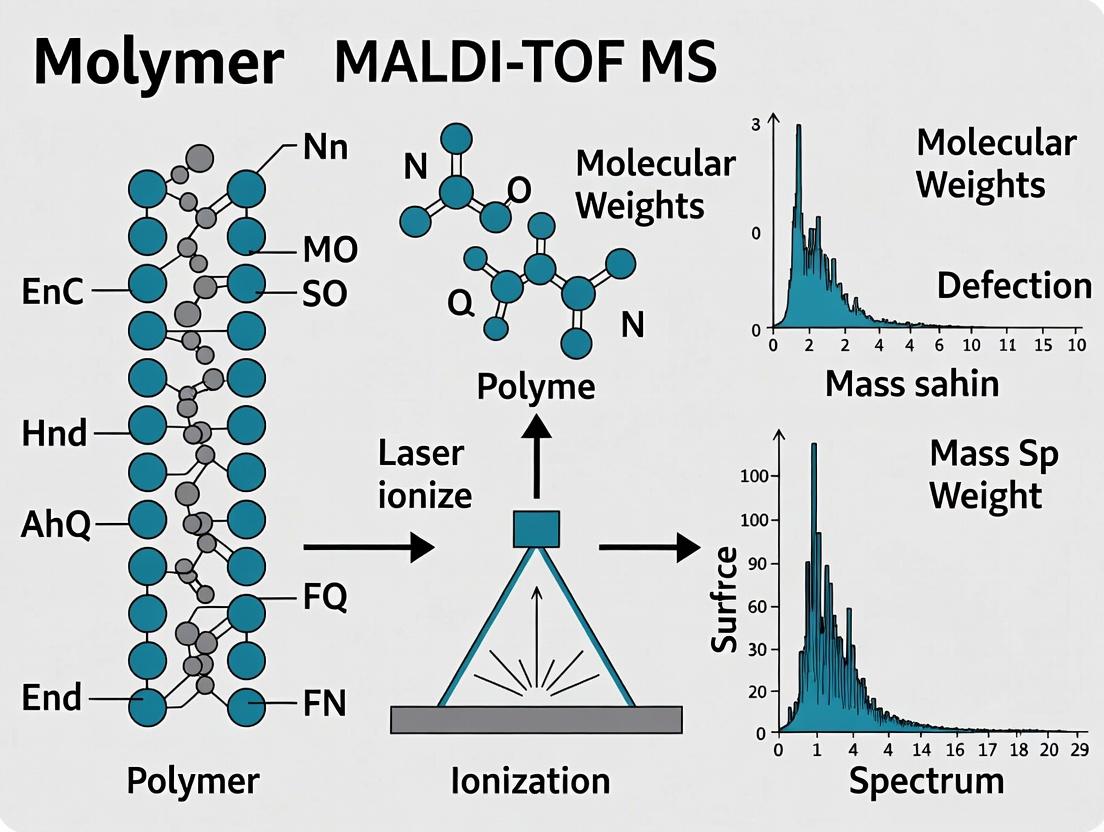
Within the context of polymer molecular weight research, Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) has emerged as an indispensable analytical technique. Its unique capacity to gently desorb and ionize large, non-volatile biomolecules and synthetic polymers, followed by precise mass separation, allows for the accurate determination of molecular weight distributions, end-group analysis, and copolymer composition. This Application Note details the fundamental principles, protocols, and critical considerations for applying MALDI-TOF MS to polymer characterization.
Core Principles: A Three-Stage Process
The MALDI-TOF MS process integrates three sequential physical events to transform a solid sample into a interpretable mass spectrum.
Desorption
Desorption is the phase transition of the analyte from the solid state into the gas phase, initiated by a pulsed ultraviolet laser (commonly a 337 nm N₂ laser). The analyte is pre-mixed with a vast molar excess of a small, UV-absorbing organic matrix (e.g., Dithranol, α-Cyano-4-hydroxycinnamic acid). Upon laser irradiation, the matrix rapidly absorbs energy, leading to explosive sublimation and the ejection of both matrix and intact, neutral analyte molecules into the expanding gas plume.
Ionization
Ionization occurs concurrently with and immediately after desorption within the hot, dense gas plume. The primary mechanism for polymers is proton transfer. In positive ion mode, the photo-excited matrix (M) donates a proton (H⁺) to the analyte (A): [M + H]⁺ + A → M + [A + H]⁺. Other adduct formations ([A+Na]⁺, [A+K]⁺) are also common, especially when trace salts are present. This "soft" ionization minimizes fragmentation, preserving the intact polymer chain.
Time-of-Flight (TOF) Separation
The generated ions are accelerated by a strong electric field (typically 20-25 kV) into a field-free drift tube. All ions gain the same kinetic energy: ½mv² = zeV, where m is mass, v is velocity, z is charge, e is the elementary charge, and V is the acceleration voltage. Lighter ions achieve higher velocities and reach the detector first. The time-of-flight (t) is directly related to the square root of the mass-to-charge ratio (m/z): t ∝ √(m/z). The detector records ion intensity versus time, which is calibrated to produce a mass spectrum.
Diagram: The Three-Stage MALDI-TOF MS Workflow
Experimental Protocol for Polymer Analysis
Materials & Sample Preparation
Successful analysis hinges on proper sample preparation. The key steps are:
Matrix Selection: Choose based on polymer compatibility and laser wavelength.
- For synthetic polymers (e.g., PS, PMMA): Dithranol, Trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB).
- For polar polymers/peptides: α-Cyano-4-hydroxycinnamic acid (CHCA), 2,5-Dihydroxybenzoic acid (DHB).
Cationization Agent: Added to promote uniform adduct formation.
- Sodium trifluoroacetate (NaTFA) or Potassium trifluoroacetate (KTFA) at ~1 mM concentration in the matrix solution.
Solvent: High purity, typically Tetrahydrofuran (THF) for non-polar polymers or Acetonitrile/Water mixtures for polar systems.
Step-by-Step Protocol: Dried-Droplet Method for Polystyrene
- Objective: Determine the molecular weight distribution of a polystyrene standard.
- Materials:
- Polystyrene analyte (e.g., 5 kDa standard).
- Matrix: DCTB (20 mg/mL in THF).
- Cationization Agent: NaTFA (10 mg/mL in THF).
- Solvent: HPLC-grade THF.
- MALDI target plate (stainless steel or ground steel).
| Step | Procedure | Critical Parameter |
|---|---|---|
| 1. Solution Prep | Prepare analyte solution at ~1-2 mg/mL in THF. Mix matrix, analyte, and salt solutions in a 10:1:1 volume ratio (e.g., 10 µL DCTB : 1 µL PS : 1 µL NaTFA). Vortex gently. | Analyte concentration must be low to avoid aggregation and ensure matrix isolation. |
| 2. Spotting | Apply 0.5 - 1.0 µL of the mixture onto the MALDI target. Allow to dry at ambient temperature, forming a homogeneous crystalline layer. | Slow, even drying produces the most consistent crystals and ion yield. |
| 3. Instrument Setup | Load target into spectrometer. Set instrument parameters: Linear positive ion mode, acceleration voltage 25 kV, delayed extraction optimized for mass range. | Delayed extraction is crucial for resolving power in linear mode. |
| 4. Data Acquisition | Use a low laser fluence (~5-10% above threshold). Acquire spectra from at least 10 different spots per sample, summing 200-500 shots per spot. | "Sweet spot" phenomenon requires sampling multiple locations. |
| 5. Calibration | Calibrate using a dedicated polymer standard (e.g., PEG or PS calibrant) spotted separately or as an internal mixture. | External calibration is acceptable for narrow dispersity polymers. |
Diagram: MALDI-TOF MS Polymer Analysis Protocol
The Scientist's Toolkit: Essential Research Reagent Solutions
| Reagent/Material | Function in MALDI-TOF MS Polymer Analysis |
|---|---|
| DCTB Matrix | A superior "universal" matrix for synthetic polymers. Its high absorption at 337 nm and good solubility in organic solvents promotes efficient desorption and homogeneous co-crystallization with hydrophobic polymers. |
| CHCA Matrix | Standard matrix for peptides and polar polymers. Facilitates proton transfer and often produces high-resolution spectra in the low to mid mass range (<20 kDa). |
| Sodium Trifluoroacetate (NaTFA) | The most common cationization agent. Provides a consistent source of Na⁺ ions to form [M+Na]⁺ adducts, simplifying the spectral pattern compared to mixed H⁺/Na⁺/K⁺ adducts. |
| THF (HPLC Grade) | Preferred solvent for many non-polar synthetic polymers (e.g., polystyrene, poly(methyl methacrylate)). Ensures complete dissolution of both analyte and matrix for homogeneous sample preparation. |
| Ground Steel Target Plate | A reusable, non-reflective target plate. Its surface promotes even crystal growth for the dried-droplet method, improving shot-to-shot reproducibility. |
| Polymer Calibration Standards | Narrow dispersity polymers (e.g., PEG, PS, PMMA) of known molar mass. Essential for external or internal instrument calibration to ensure accurate mass assignment. |
Data Interpretation and Key Metrics for Polymers
The resulting spectrum displays a series of peaks corresponding to individual oligomers differing by one repeat unit mass. Quantitative analysis yields critical polymer parameters:
| Parameter | Formula (from MALDI Data) | Significance |
|---|---|---|
| Number-Average Molecular Weight (Mₙ) | Σ(Nᵢ * Mᵢ) / ΣNᵢ | The arithmetic mean mass, sensitive to low molecular weight species. |
| Weight-Average Molecular Weight (M_w) | Σ(Nᵢ * Mᵢ²) / Σ(Nᵢ * Mᵢ) | The mass-weighted mean, sensitive to high molecular weight species. |
| Dispersity (Đ) | M_w / Mₙ | Measure of polymer chain length distribution. Đ=1 is monodisperse. |
Note: While MALDI provides excellent accuracy for Mₙ and Đ for polymers with Đ < ~1.2, its sensitivity can be biased against very high mass chains, potentially skewing M_w. This must be considered in the broader thesis context.
Why MALDI-TOF for Polymers? Advantages Over Traditional Techniques like SEC/GPC
Application Notes
Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) has emerged as a powerful technique for polymer analysis, offering unique advantages over traditional methods like Size Exclusion Chromatography (SEC), also known as Gel Permeation Chromatography (GPC). Within a thesis on polymer molecular weight research, MALDI-TOF MS provides complementary and often more definitive data.
Key Advantages of MALDI-TOF MS:
- Absolute Molecular Weights: Unlike SEC/GPC, which relies on calibration with polymer standards, MALDI provides absolute molecular weights (Mn, Mw) directly from the mass spectrum, eliminating calibration errors.
- Structural and End-Group Analysis: MALDI-TOF MS yields detailed information on polymer microstructure, including end-group identification, repeat unit confirmation, and detection of side reactions or cyclic species, which are typically inaccessible via SEC.
- High Resolution for Low Mass Polymers: It excels at characterizing low to medium molecular weight polymers (typically up to ~100 kDa) with exceptional mass resolution, revealing individual oligomers.
- Speed and Sample Throughput: Analysis time per sample is rapid (minutes), and the technique is amenable to high-throughput screening when combined with automated target plates.
- Polydispersity Index (PDI) from Direct Measurement: The PDI is calculated from the absolute molecular weight distribution of detected oligomers, not inferred from a chromatogram's breadth.
Limitations and Complementary Role: MALDI-TOF MS can struggle with high molecular weight polymers (>100-200 kDa) and samples with very high polydispersity (PDI > 1.2-1.3), where SEC/GPC remains superior. It is also matrix-dependent and requires careful sample preparation. Therefore, the most robust polymer characterization strategy often integrates both techniques: SEC for bulk, high-MW distributions, and MALDI for detailed structural insight and absolute calibration of SEC columns.
Quantitative Comparison: MALDI-TOF MS vs. SEC/GPC
Table 1: Core Technical Comparison
| Feature | MALDI-TOF MS | SEC/GPC |
|---|---|---|
| Molecular Weight Type | Absolute (from m/z) | Relative (vs. calibration standards) |
| Mass Range (optimal) | Up to ~100-200 kDa | Up to 10+ MDa (aqueous), ~2 MDa (organic) |
| Information Obtained | Mn, Mw, PDI, end-group, repeat unit, copolymer sequence, impurities | Mn, Mw, Mz, PDI (bulk averages) |
| Sample Throughput | High (minutes/sample) | Medium (20-30 mins/sample) |
| Polydispersity Limitation | Challenging for PDI > ~1.3 | Handles broad distributions (PDI > 2) |
| Solvent Compatibility | Analyzed solid-state | Requires specific eluents for dissolution |
| Key Limitation | Mass discrimination, matrix effects, poor for very high MW | Calibration uncertainty, band broadening, no structural data |
Table 2: Typical Data Quality Metrics for a Polystyrene Standard (PS 2,500 Da)
| Parameter | MALDI-TOF MS Result | SEC/GPC Result (PS-calibrated) | Reference/True Value |
|---|---|---|---|
| Number-Avg. MW (Mn) | 2,480 Da | 2,550 Da | 2,500 Da |
| Weight-Avg. MW (Mw) | 2,510 Da | 2,610 Da | 2,550 Da |
| Polydispersity Index (PDI) | 1.012 | 1.024 | 1.020 |
| Additional Data | End-group identified as H+/Na+, Individual oligomers resolved | Chromatogram peak only | N/A |
Experimental Protocols
Protocol 1: Standard Sample Preparation for Synthetic Polymers (e.g., Polystyrene, PMMA)
This "dried droplet" method is a common starting point for hydrophobic polymers.
Research Reagent Solutions & Essential Materials:
| Item | Function |
|---|---|
| Matrix (e.g., DCTB, trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile) | Absorbs laser energy, facilitates desorption/ionization of analyte with minimal fragmentation. |
| Cationization Agent (e.g., NaTFA, Sodium Trifluoroacetate) | Provides cations (Na+, K+, Ag+) to ionize neutral polymer chains for detection. |
| Solvent (e.g., HPLC-grade THF, Toluene, Chloroform) | Dissolves polymer, matrix, and salt to form a homogeneous solution. |
| MALDI Target Plate (Stainless steel or reusable) | Platform for sample crystallization. |
| Calibration Standard (e.g., PPG, Polystyrene narrow standards) | For external or internal instrument mass axis calibration. |
| Microcentrifuge Tubes & Pipettes | For sample mixing and precise liquid handling. |
Detailed Methodology:
- Solution Preparation:
- Prepare matrix solution: Dissolve DCTB in THF at a concentration of 20-30 mg/mL.
- Prepare cationization solution: Dissolve NaTFA in THF at 1-10 mg/mL.
- Prepare analyte solution: Dissolve polymer in THF at ~1-5 mg/mL.
- Mixing: In a microcentrifuge tube, mix the solutions in a volume ratio of 10:1:1 (Matrix : Analyte : Salt). Vortex gently.
- Spotting: Pipette 0.5-1.0 µL of the mixture onto a clean MALDI target plate. Allow to dry at room temperature, forming a homogeneous crystalline layer.
- Calibration: Spot a calibration standard (e.g., PPG 1000-2500) separately or mixed internally with the analyte.
- Instrument Operation: Load target into vacuum chamber. Acquire spectra in reflection positive ion mode. Adjust laser power to achieve optimal signal-to-noise without inducing fragmentation.
Protocol 2: Sample Preparation for Polydisperse or Challenging Polymers
For broader distributions or polymers prone to discrimination.
Methodology:
- Fractionation: Pre-fractionate the polymer sample using a quick SEC or LC-Transform system to collect narrower MW fractions. Analyze each fraction by MALDI-TOF MS separately.
- Layered/Sandwich Method: To reduce mass bias:
- First, spot a thin layer of matrix solution and let it dry.
- Then, spot the analyte/salt mixture on top of the pre-coated matrix layer.
- Finally, spot another thin layer of matrix solution on top.
- Matrix Selection: Experiment with alternative matrices (e.g., Dithranol for PEG, Sinapinic Acid for higher MW proteins/polymers) and cations (e.g., Ag+ for polyolefins, K+ for PEG).
- Data Merging: For fractionated samples, recombine the MW distributions from each MALDI spectrum mathematically to reconstruct the full distribution.
Visualizations
Title: Polymer Thesis Method Integration Map
Title: MALDI-TOF Polymer Analysis Workflow
Within the context of developing a robust MALDI-TOF MS method for polymer characterization, the accurate determination of molecular weight averages and dispersity is paramount. These key outputs—Mₙ, Mᵥ, and Đ—provide fundamental insights into polymer synthesis efficacy, batch-to-batch consistency, and ultimately, the material's physical properties and performance in applications such as drug delivery systems and biodegradable implants. This application note details the protocols for obtaining these critical parameters using MALDI-TOF MS, emphasizing calibration, sample preparation, and data analysis tailored for synthetic polymer research.
Molecular weight distributions are characterized by different averages. The Number-Average Molecular Weight (Mₙ) is sensitive to the total number of polymer chains, while the Weight-Average Molecular Weight (Mᵥ) is influenced by the mass of the chains. Dispersity (Đ = Mᵥ/Mₙ) describes the breadth of the distribution.
Table 1: Molecular Weight Averages and Dispersity Definitions
| Parameter | Mathematical Definition | Sensitivity |
|---|---|---|
| Number-Average (Mₙ) | Mₙ = Σ(NᵢMᵢ) / ΣNᵢ | Number of molecules |
| Weight-Average (Mᵥ) | Mᵥ = Σ(NᵢMᵢ²) / Σ(NᵢMᵢ) | Mass of molecules |
| Dispersity (Đ) | Đ = Mᵥ / Mₙ | Polydispersity index (PDI) |
Table 2: Typical Đ Values and Polymer Characterization
| Dispersity (Đ) Range | Typical Polymer Synthesis Method | Implication for Properties |
|---|---|---|
| 1.01 – 1.10 | Living anionic polymerization, precise CRP | Very narrow distribution, uniform properties |
| 1.1 – 1.5 | Well-controlled ATRP, RAFT | Controlled distribution |
| 1.5 – 2.0 | Conventional radical polymerization | Moderate distribution |
| >2.0 | Step-growth polymerization, poorly controlled reactions | Broad distribution, batch variability |
Experimental Protocol: MALDI-TOF MS for Mₙ, Mᵥ, and Đ Determination
Materials and Reagent Solutions
Table 3: Research Reagent Toolkit for Polymer MALDI-TOF MS
| Item | Function & Brief Explanation |
|---|---|
| MALDI Matrix (e.g., DCTB, CHCA) | Absorbs laser energy, promotes soft ionization of the analyte. DCTB is often preferred for polymers. |
| Cationizing Agent (e.g., NaTFA, KTFA) | Promotes the formation of [M+Cat]⁺ ions (Cat = Na⁺, K⁺, Ag⁺) for consistent detection. |
| HPLC-grade Solvents (THF, CHCl₃, ACN) | Used for dissolving polymer and matrix to ensure purity and prevent interference. |
| Polystyrene or PEG Calibration Standards | Narrow dispersity polymers with known Mₙ for external instrument calibration. |
| Stainless Steel or Gold-plated MALDI Target Plate | Sample deposition surface. |
| Precision Micropipettes (0.5-10 µL) | For accurate and reproducible sample/matrix spotting. |
Detailed Step-by-Step Protocol
Step 1: Sample and Matrix Preparation
- Prepare a polymer solution (~1-10 mg/mL) in a suitable solvent (e.g., THF for polystyrenes).
- Prepare a matrix solution (~20 mg/mL) in the same solvent. DCTB is recommended for broad polymer compatibility.
- Prepare a cationizing agent solution (~10 mg/mL).
- Mix solutions in a vial at a typical volume ratio of Polymer : Matrix : Salt = 1 : 10 : 1.
- Vortex thoroughly for 30 seconds.
Step 2: Target Spotting and Sample Crystallization
- Using a micropipette, deposit 0.5-2.0 µL of the mixture onto a clean MALDI target plate.
- Allow the spot to dry under ambient conditions, forming a homogeneous crystalline layer.
- For difficult samples, employ the "dried droplet" or "layer" method for improved homogeneity.
Step 3: Instrument Calibration
- Spot a calibration standard (e.g., PEG 2000) using an identical protocol.
- Acquire a spectrum of the standard.
- Perform external calibration by identifying at least 5-10 known [M+Cat]⁺ peaks in the calibration standard's spectrum to generate a calibration curve.
Step 4: Data Acquisition
- Insert the target plate into the MALDI-TOF mass spectrometer.
- Set instrument parameters in linear positive ion mode for higher mass polymers to obtain a resolved envelope of peaks. Reflector mode can be used for lower masses (<10 kDa) for isotopic resolution.
- Key parameters: Laser intensity (just above threshold), acceleration voltage, and detector gain optimized for signal-to-noise.
- Acquire spectra from multiple positions across each sample spot to account for heterogeneity.
Step 5: Data Processing and Calculation of Mₙ, Mᵥ, and Đ
- Spectrum Pre-treatment: Apply baseline correction and smoothing if necessary.
- Peak Assignment: Identify the series of peaks corresponding to [M+Cat]⁺ ions, accounting for the polymer's repeat unit mass (e.g., 104 Da for styrene, 44 Da for ethylene glycol).
- Intensity Extraction: For each peak in the oligomeric series (i), record the mass-to-charge ratio (m/z)ᵢ and the corresponding absolute signal intensity (I)ᵢ. Note: In linear MALDI-TOF of polymers, signal intensity is assumed proportional to the relative abundance by weight (wᵢ).
- Conversion to Number Distribution:
- Calculate the relative weight abundance: wᵢ = Iᵢ / ΣIᵢ.
- Calculate the number of moles for each species: nᵢ = wᵢ / Mᵢ.
- Calculate the relative number abundance: Nᵢ = nᵢ / Σnᵢ.
- Calculation of Averages:
- Mₙ = Σ(Nᵢ * Mᵢ)
- Mᵥ = Σ(wᵢ * Mᵢ)
- Đ = Mᵥ / Mₙ
- Validation: Compare results with known standards. Ensure the mass range captured in the spectrum is representative of the entire polymer distribution.
Visualization of Workflows and Relationships
Title: MALDI-TOF MS Workflow for Polymer Mₙ, Mᵥ, and Đ
Title: Relationship Between Mₙ, Mᵥ, and Đ
1. Introduction Within the broader thesis on MALDI-TOF MS for polymer molecular weight research, end-group analysis emerges as a critical, non-negotiable parameter for accurate characterization. While bulk molecular weight (Mn, Mw) provides foundational data, it is the identification and quantification of polymer chain termini that enable precise determination of polymerization mechanisms, kinetics, and final polymer structure. This is especially vital in drug development for polymeric excipients, conjugates, and controlled-release formulations, where end-groups dictate stability, bioactivity, and biodistribution. MALDI-TOF MS, with its soft ionization and mass accuracy, is the premier technique for direct end-group analysis by measuring the exact mass of individual oligomers.
2. Quantitative Data Summary: End-Group Impact on Polymer Properties
Table 1: Influence of End-Group Chemistry on Key Polymer Properties
| Polymer Type | End-Group A | End-Group B | Key Property Affected | Quantitative Change | Analytical Method |
|---|---|---|---|---|---|
| Poly(ethylene glycol) (PEG) | -OH (Hydroxyl) | -OCH3 (Methoxy) | Plasma Circulation Half-life | Increase from ~2 hr to ~20 hr (in mice) | SEC-MALS, in vivo PK |
| Poly(lactic-co-glycolic acid) (PLGA) | -COOH (Carboxyl) | -CH3 (Ester) | Degradation Rate (pH 7.4) | Acceleration by ~15-20% | GPC, pH-stat |
| Polystyrene (PS) | -H (Hydrogen) | -Br (Bromine) | Chain Extension Feasibility | >95% re-initiation efficiency | NMR, MALDI-TOF MS |
| Polycaprolactone (PCL) | -OH | -N3 (Azide) | Subsequent Bioconjugation Yield | Coupling yield >98% via click chemistry | UV-Vis, HPLC |
Table 2: MALDI-TOF MS Resolving Power Requirements for Common Polymers
| Polymer | Repeat Unit Mass (Da) | Typical End-Group Mass Difference (Da) | Required MS Resolution (m/Δm) | Recommended MALDI Matrix |
|---|---|---|---|---|
| Poly(methyl methacrylate) (PMMA) | 100.05 | 1.01 (H vs. CH3) | ~10,000 | DCTB |
| Polyethylene glycol (PEG) | 44.03 | 15.99 (OH vs. OCH3) | ~3,000 | DHB |
| Polystyrene (PS) | 104.06 | 78.92 (H vs. Br) | ~1,500 | DCTB |
| Nylon-6 | 113.08 | 22.03 (NH2 vs. COOH) | ~5,000 | HFIP-doped DHB |
3. Detailed Experimental Protocols
Protocol 3.1: Sample Preparation for MALDI-TOF MS End-Group Analysis Objective: To prepare a polymer sample for precise end-group identification via MALDI-TOF MS. Materials: Polymer sample, MALDI matrix (e.g., DCTB, DHB), cationization salt (e.g., NaTFA, KTFA), THF or chloroform (HPLC grade), methanol, MALDI target plate. Procedure:
- Prepare a 10 mg/mL stock solution of the polymer in a suitable solvent (e.g., THF for PS, chloroform for PEG).
- Prepare a 20 mg/mL solution of the matrix in the same solvent.
- Prepare a 10 mg/mL solution of the cationization salt (e.g., NaTFA) in methanol.
- Mix the solutions in a vial at a volume ratio of Polymer:Matrix:Salt = 5:10:1.
- Spot 0.5-1.0 µL of the final mixture onto the MALDI target plate and allow to dry under ambient conditions to form homogeneous crystals.
- Insert the plate into the MALDI-TOF mass spectrometer.
Protocol 3.2: Data Processing and End-Group Calculation from MALDI Spectrum Objective: To derive end-group masses and identify chemical structures from the isotopic oligomer series. Software Requirements: MALDI control/analysis software (e.g., Bruker flexAnalysis, Shimadzu MALDI Solutions). Procedure:
- Acquire spectrum in reflection positive ion mode. Calibrate using a known polymer standard (e.g., PEG 2000).
- Identify the isotopic pattern of a single oligomer peak. Determine the monoisotopic mass (M_mono).
- For a given oligomer with 'n' repeat units, the mass is described by: M_mono = [M(End-Group A) + M(End-Group B) + n * M(Repeat Unit) + M(Cation)]⁺
- Select two adjacent peaks in the series (differing by one repeat unit, n and n+1).
- Solve the simultaneous equations:
- Peakn = EndA + EndB + n*Repeat + Cation
- Peak(n+1) = EndA + EndB + (n+1)*Repeat + Cation
- Subtract the two equations to calculate the exact mass of the repeat unit: M(Repeat) = Peak(n+1) - Peakn.
- Substitute M(Repeat) back into the first equation to calculate the combined end-group mass: M(EndA) + M(EndB) = Peak_n - n*M(Repeat) - M(Cation).
- Compare the combined end-group mass to a database of potential initiator/terminator masses to propose chemical structures.
4. Visualizations
5. The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in End-Group Analysis | Example Product/Chemical |
|---|---|---|
| MALDI Matrices | Absorbs laser energy, facilitates soft ionization of polymer analyte with minimal fragmentation. | Trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB), 2,5-Dihydroxybenzoic acid (DHB) |
| Cationization Salts | Provides cations (Na⁺, K⁺, Ag⁺) to promote ionization of non-polar polymers by forming [M+Cation]⁺ adducts. | Sodium trifluoroacetate (NaTFA), Potassium trifluoroacetate (KTFA), Silver trifluoroacetate (AgTFA) |
| Polymer Standards | Essential for instrumental mass calibration and validating resolution/accuracy for specific polymer classes. | Narrow dispersity PEG, PS, PMMA standards with defined end-groups. |
| Ultra-Pure Solvents | Prevents contamination and unwanted adduct formation that can complicate mass spectra interpretation. | HPLC-grade Tetrahydrofuran (THF), Chloroform, Methanol. |
| Functionalized End-Group Standards | Used as internal references or for method development to confirm identification of specific terminal moieties. | α-Methoxy-ω-hydroxy PEG, Bromo-terminated Polystyrene, Azide-PEG-NHS Ester. |
In the broader context of developing a robust MALDI-TOF MS methodology for polymer molecular weight and dispersity (Ð) analysis, the selection and optimization of core components are critical. The interplay between the matrix, cationization agent, solvent system, and substrate dictates the quality of the desorption/ionization process, directly impacting mass spectral resolution, signal intensity, and the accuracy of molecular weight determination. This document provides detailed application notes and protocols centered on these four essential components, with a focus on synthetic polymers.
The Scientist's Toolkit: Research Reagent Solutions
Table 1: Essential Materials for MALDI-TOF MS Analysis of Polymers
| Component | Example Reagents/Solutions | Primary Function | Key Consideration |
|---|---|---|---|
| Matrix | Dithranol (1,8,9-anthracentriol), α-Cyano-4-hydroxycinnamic acid (CHCA), 2,5-Dihydroxybenzoic acid (DHB) | Absorbs laser energy, facilitates polymer desorption/ionization, and reduces polymer degradation. | Must have strong UV absorption at the laser wavelength (usually 337 nm) and good co-crystallization with the analyte. |
| Cationization Agent | Sodium trifluoroacetate (NaTFA), Potassium trifluoroacetate (KTFA), Silver trifluoroacetate (AgTFA) | Provides cations (Na⁺, K⁺, Ag⁺) to adduct to polymer chains, enabling ionization and detection in positive ion mode. | Choice depends on polymer affinity; Ag⁺ is often used for polymers with π-systems (e.g., polystyrene). |
| Solvent System | Tetrahydrofuran (THF), Chloroform, Acetone, Methanol, Water | Dissolves all sample components (polymer, matrix, salt) to form a homogeneous solution for uniform co-crystallization. | Must be volatile for rapid drying. A ternary mixture (e.g., THF/acetone/water) often improves crystal morphology. |
| Substrate | Polished Steel MALDI Target Plates, Ground Steel Plates with Hydrophobic Coating | Platform for sample deposition. Surface properties influence droplet spreading and final crystal size/morphology. | Hydrophobic coatings can restrict spot size, leading to more concentrated sample spots. |
Component Optimization & Protocols
Matrix Selection and Preparation Protocol
Objective: To prepare a saturated matrix solution for optimal co-crystallization with the polymer analyte. Protocol for Dithranol (for Polystyrene, PMMA):
- Weigh 20 mg of dithranol into a 1 mL HPLC vial.
- Add 1 mL of HPLC-grade THF. Cap and vortex for 1 minute.
- Sonicate for 5 minutes to ensure full saturation. The solution may have undissolved solid.
- Centrifuge briefly (30 sec, 5000 rpm) to pellet any particulate matter.
- Use the supernatant for sample preparation. Store at room temperature, protected from light, for up to 1 week.
Table 2: Common Matrices for Polymer Analysis
| Matrix | λ_max (nm) | Ideal Polymer Types | Typical Conc. (mg/mL) | Primary Solvent |
|---|---|---|---|---|
| Dithranol | ~340 | Polystyrene, Polyesters, PMMA | 20 | THF |
| CHCA | ~350 | Peptides, Polar Polymers | 10 | Acetonitrile/0.1% TFA (1:1) |
| DHB | ~340 | Polysaccharides, PEG | 20 | Acetone/Methanol (9:1) |
| trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB) | ~340 | Broad applicability, less fragmentation | 20 | THF |
Cationization Agent Optimization Protocol
Objective: To determine the optimal cationization agent and concentration for a given polymer. Experimental Procedure:
- Prepare a stock solution of the polymer (e.g., PEG 2000) at 10 mg/mL in THF.
- Prepare stock solutions of cationization agents (NaTFA, KTFA, AgTFA) at 10 mg/mL in THF or methanol.
- Prepare the matrix solution (e.g., DHB for PEG) at 20 mg/mL in THF.
- Prepare three sample mixtures in separate vials with a final volume of 20 µL each:
- Vial 1 (Na⁺): 10 µL polymer, 5 µL matrix, 5 µL NaTFA stock.
- Vial 2 (K⁺): 10 µL polymer, 5 µL matrix, 5 µL KTFA stock.
- Vial 3 (Ag⁺): 10 µL polymer, 5 µL matrix, 5 µL AgTFA stock.
- Vortex each mixture for 10 seconds.
- Spot 0.5-1 µL of each mixture onto the MALDI target plate and allow to dry in ambient air.
- Acquire MALDI-TOF spectra for each spot using identical instrument settings (laser power, detector gain).
Table 3: Cationization Agent Selection Guide
| Polymer Type | Recommended Cation | Typical Molar Ratio (Polymer Unit:Salt) | Observed Adduct |
|---|---|---|---|
| Poly(ethylene glycol) (PEG) | Na⁺ or K⁺ | 1:1 to 1:5 | [M+Na]⁺ or [M+K]⁺ |
| Polystyrene (PS) | Ag⁺ | 1:2 to 1:10 | [M+Ag]⁺ |
| Poly(methyl methacrylate) (PMMA) | Na⁺ or Li⁺ | 1:2 to 1:5 | [M+Na]⁺ or [M+Li]⁺ |
| Polyesters (e.g., PLA) | Na⁺ | 1:2 to 1:5 | [M+Na]⁺ |
Solvent System & Sample Spotting Protocol
Objective: To deposit a homogeneous, microcrystalline sample spot for reproducible analysis. Ternary Solvent Method for Hydrophobic Polymers:
- Prepare a working sample solution by mixing in this order in an HPLC vial:
- Polymer Solution: 10 µL of polymer (1-10 mg/mL in THF).
- Matrix Solution: 10 µL of matrix (e.g., 20 mg/mL DCTB in THF).
- Salt Solution: 2 µL of cationization agent (e.g., 10 mg/mL AgTFA in THF).
- Ternary Additive: 3 µL of a polar, volatile solvent (e.g., acetone) to modify crystallization kinetics.
- Vortex the mixture for 15-30 seconds until fully homogeneous.
- Using a calibrated pipette, deposit 0.5 - 1.0 µL of the mixture onto a polished steel MALDI target.
- Allow the spot to dry slowly at ambient temperature and humidity. For more controlled drying, place the target in a desiccator for 2-5 minutes.
- Visually inspect the spot under a microscope if available. A homogeneous, microcrystalline "frosted" appearance is ideal.
Substrate Pre-Treatment Protocol
Objective: To clean and/or functionalize the target plate to improve sample adhesion and spot homogeneity. Protocol for Cleaning Polished Steel Targets:
- Sonicate the target plate in HPLC-grade methanol for 5 minutes.
- Transfer the plate and sonicate in HPLC-grade acetone for 5 minutes.
- Rinse the plate thoroughly with isopropanol.
- Dry the plate under a stream of dry, oil-free nitrogen gas.
- Use immediately or store in a clean desiccator.
Visualizing the MALDI-TOF MS Workflow for Polymers
Title: MALDI-TOF MS Polymer Analysis Workflow
The Role of Components in the Desorption/Ionization Pathway
Title: MALDI Desorption/Ionization Mechanism & Component Roles
Step-by-Step MALDI-TOF MS Protocol: Sample Preparation, Data Acquisition, and Analysis for Diverse Polymers
Introduction This application note is framed within a broader thesis on developing robust MALDI-TOF MS methodologies for polymer molecular weight and structural analysis. Matrix selection is a critical parameter influencing ionization efficiency, spectral quality, and quantitative accuracy. This guide details the application of four common matrices—trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB), 2,5-Dihydroxybenzoic acid (DHB), α-Cyano-4-hydroxycinnamic acid (CHCA), and Sinapinic acid (SA)—across major polymer classes, supported by current experimental data and protocols.
1. Matrix-Polymer Compatibility: Quantitative Performance Summary The following table consolidates key performance metrics for each matrix-polymer combination based on recent literature and empirical data. Abbreviations: PS (Polystyrene), PEG/PEO (Polyethylene glycol/oxide), PMMA (Poly(methyl methacrylate)), Polyesters (e.g., PLA, PCL), Polycarbonates.
| Matrix | Optimal Polymer Classes | Typical [M+Na]+/% | Typical [M+Ag]+/% | Key Advantage | Major Limitation |
|---|---|---|---|---|---|
| DCTB | PS, Polybutadiene, Polycarbonates, Polyesters | 10-25% (with Ag) | 60-85% (primary ion) | Low fragmentation, excellent for hydrocarbons; works with Ag salt doping. | Poor for polar polymers without cationization agent. |
| DHB | PEG, Polysaccharides, Polyethers, Polyacrylates | 70-90% (with Na/K) | N/A | Good for polar polymers; forms fine crystals. | "Sweet spot" phenomenon; heterogeneous crystallization. |
| CHCA | Low-Mw PEG, Oligomers, Polyamides (<10 kDa) | 50-80% (with Na/K) | N/A | Fast crystallization, high sensitivity for low-Mw. | In-source decay for polymers >~10 kDa. |
| SA | Proteins, Synthetic Polypeptides, PMMA, High-Mw PEG (>10 kDa) | 60-85% (with Na/K) | N/A | Gentle ionization for high-Mw, less fragmentation. | Can form multiple adduct species. |
2. Detailed Experimental Protocols
Protocol 1: General Sample Preparation for Synthetic Polymers (Dried-Droplet Method) Objective: To prepare a homogeneous matrix:analyze:salt mixture for MALDI-TOF MS analysis. Materials: MALDI matrix (DCTB, DHB, CHCA, or SA), polymer sample, cationization salt (e.g., NaTFA, KCL AgTFA), suitable solvent (THF, acetone, chloroform, ACN, water). Procedure:
- Prepare matrix solution (20 mg/mL) and cationization salt solution (10 mg/mL) in appropriate solvents (e.g., THF for DCTB; 50:50 ACN:H₂O for DHB).
- Prepare polymer sample solution at ~1-2 mg/mL in a solvent compatible with the matrix solution.
- Mix solutions in a vial at typical volume ratios: Matrix : Polymer : Salt = 10 : 1 : 1 (v/v/v). Vortex briefly.
- Pipette 0.5-1.0 µL of the mixture onto a MALDI target spot.
- Allow to air dry at ambient temperature to form a crystalline deposit.
- Insert target into mass spectrometer for analysis.
Protocol 2: DCTB with Silver Cationization for Aromatic/Aliphatic Polymers Objective: To analyze non-polar polymers (e.g., PS, polybutadiene) via efficient Ag⁺ adduct formation. Specific Materials: DCTB matrix, AgTFA salt, THF (HPLC grade), polymer sample. Procedure:
- Dissolve DCTB (20 mg/mL) and AgTFA (10 mg/mL) separately in THF. Sonicate if needed.
- Dissolve polymer sample in THF at ~2 mg/mL.
- Combine 10 µL DCTB, 1 µL polymer, and 1 µL AgTFA solution. Mix thoroughly.
- Spot 1 µL on target. Dry in darkness (Ag salts are light-sensitive).
- Acquire spectra in positive linear or reflection mode.
Protocol 3: DHB for Polyethers and Polar Polymers Objective: To achieve uniform sample crystallization for polar polymers like PEG. Specific Materials: DHB matrix, NaTFA salt, Acetonitrile (ACN), Deionized Water. Procedure:
- Prepare a saturated solution of DHB in 50:50 (v/v) ACN:H₂O. This may be >30 mg/mL.
- Prepare polymer solution (1 mg/mL) in a compatible solvent (e.g., water, MeOH).
- Prepare NaTFA solution (10 mg/mL) in water or MeOH.
- Mix 10 µL DHB, 2 µL polymer, and 1 µL NaTFA.
- Spot 1 µL on target and allow to dry slowly under ambient conditions to promote larger crystal formation.
- For best results, manually locate "sweet spots" (areas of homogeneous co-crystallization) on the target for laser irradiation.
3. Visualization: Matrix Selection and Workflow
Title: Decision Workflow for Polymer Matrix Selection
4. The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function/Benefit | Example/Note |
|---|---|---|
| DCTB Matrix | Superior for non-polar polymers via Ag⁺ cationization; minimizes fragmentation. | Use with AgTFA for PS, polyolefins. Light-sensitive. |
| DHB Matrix | Standard for polar polymers (PEG, PMMA); promotes [M+Alkali]+ ions. | Use slow drying for larger crystals; locate "sweet spots". |
| CHCA Matrix | High sensitivity for low-Mw polymers/oligomers; fast crystallization. | Can cause in-source decay for larger polymers. |
| SA Matrix | "Soft" matrix for higher mass polymers (>10kDa) and biopolymers. | Common for proteins and synthetic polypeptides. |
| Silver Trifluoroacetate (AgTFA) | Primary cationization agent for polymers with low alkali affinity. | Essential for DCTB analyses of hydrocarbons. |
| Sodium/Potassium Trifluoroacetate (NaTFA/KTFA) | Standard alkali cationization salts for polar polymers. | Promotes [M+Na]+ or [M+K]+ adduct formation. |
| Tetrahydrofuran (THF, anhydrous) | Universal solvent for matrices and many synthetic polymers. | Ideal for DCTB and organic-soluble polymers. |
| Acetonitrile (ACN) & Water Mixtures | Standard solvent system for DHB, CHCA, SA and water-soluble polymers. | Common ratio is 1:1 for matrix preparation. |
| Pre-polished MALDI Target Plates | Stainless steel or gold-coated plates for sample deposition. | Ensure compatibility with instrument stage. |
| Calibration Standard Kit | Polymer or protein standards for accurate mass/axis calibration. | Use narrow dispersity PEG or PS close to sample Mw. |
Within the broader thesis investigating the MALDI-TOF MS method for polymer molecular weight and structural research, optimizing the cationization agent is a critical experimental parameter. The choice of cation (Na+, K+, Ag+, Cs+) directly influences ionization efficiency, adduct formation stability, signal-to-noise ratio, and ultimately, the accuracy and resolution of the obtained mass spectra. These factors are paramount for determining absolute molecular weights, elucidating end-group structures, and assessing polymer polydispersity—key objectives in polymer science and drug delivery system development.
Cationization Agent Properties and Selection Guide
Cationization in MALDI-TOF MS for polymers involves the addition of a metal cation to the neutral polymer chain, forming a detectable [M + Cat]+ ion. The efficacy of a cation depends on its interaction with the polymer's functional groups (e.g., ethers, esters, double bonds).
Table 1: Physicochemical Properties of Common Cationization Agents
| Cation | Ionic Radius (Å) | Electronegativity | Common Salt Forms | Affinity for Polymer Types |
|---|---|---|---|---|
| Sodium (Na+) | 1.02 | 0.93 | NaTFA, NaCl | Universal; good for polyethers, polyesters, polyacrylates. |
| Potassium (K+) | 1.38 | 0.82 | KTFA, KCl | Polyethers (higher mass resolution than Na+), polysaccharides. |
| Silver (Ag+) | 1.15 | 1.93 | AgTFA, AgNO3 | Hydrocarbon polymers (polyolefins, polystyrenes), dienes, aromatics. |
| Cesium (Cs+) | 1.67 | 0.79 | CsI, CsTFA | High-mass polymers; reduces multiple adducts, simplifies spectrum. |
Table 2: Observed Performance in MALDI-TOF MS of Poly(methyl methacrylate) (PMMA 5000 Da)
| Cation | Primary Adduct | Signal Intensity (a.u.) | Signal-to-Noise Ratio | Observed Mass Accuracy (ppm) | Tendency for Multiple Adducts |
|---|---|---|---|---|---|
| Na+ | [M+Na]+ | 1.00 (Reference) | 150 | ± 250 | Moderate |
| K+ | [M+K]+ | 0.85 | 120 | ± 300 | High ([M+Na]+ often present) |
| Ag+ | [M+Ag]+ | 1.30 | 180 | ± 200 | Low |
| Cs+ | [M+Cs]+ | 0.70 | 95 | ± 350 | Very Low |
Experimental Protocols
Protocol 1: General Method for Cationization Agent Screening
Objective: To empirically determine the optimal cationization agent for an unknown polymer sample. Materials: See "The Scientist's Toolkit" below. Procedure:
- Polymer Solution: Prepare a 10 mg/mL stock solution of the polymer in a suitable solvent (e.g., THF, chloroform, toluene).
- Matrix Solution: Prepare a 20 mg/mL solution of the matrix (e.g., DCTB, Dithranol) in the same solvent.
- Cation Salt Solutions: Prepare 10 mM solutions of each cation salt (NaTFA, KTFA, AgTFA, CsI) in a polar solvent (e.g., methanol, ethanol, or THF if compatible).
- Sample Spotting: For each cation, mix in a microcentrifuge tube:
- 10 µL polymer stock solution
- 20 µL matrix solution
- 2 µL cation salt solution Vortex briefly.
- Target Deposition: Apply 1 µL of the mixture directly to the MALDI target plate and allow to dry under ambient conditions.
- MALDI-TOF MS Analysis: Acquire spectra in positive linear or reflector mode, as appropriate for the mass range. Use consistent laser power and acquisition settings across all samples.
- Data Analysis: Compare spectra for signal intensity, signal-to-noise, adduct clarity (presence of single [M+Cat]+ peak series), and mass accuracy.
Protocol 2: Optimized Protocol for Hydrocarbon Polymers using Ag+
Objective: Achieve maximum ionization efficiency for non-polar polymers like polystyrene (PS) or polybutadiene. Materials: DCTB matrix, AgTFA salt, THF solvent. Procedure:
- Prepare polymer solution at 5 mg/mL in toluene.
- Prepare DCTB matrix solution at 30 mg/mL in THF.
- Prepare AgTFA cation solution at 2 mg/mL in THF.
- Mix in a 1:10:1 volume ratio (Polymer : Matrix : Cation). The high matrix-to-analyte ratio is critical for efficient energy transfer.
- Spot 0.5-1 µL on the target and let crystallize.
- Analyze in positive linear mode. Expect dominant [M+Ag]+ peaks with minimal fragmentation.
Diagrams and Workflows
Title: Cation Selection Decision Workflow for Polymers
Title: Cation Attachment in MALDI Ionization Process
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for Cationization Optimization Experiments
| Reagent/Material | Typical Specification/Concentration | Function in Experiment |
|---|---|---|
| MALDI Matrices | ||
| trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB) | ≥99%, 20-30 mg/mL in THF | Universal matrix, especially effective for non-polar polymers with Ag+. |
| Dithranol (1,8,9-Anthracenetriol) | ≥98%, 20 mg/mL in THF | Common for synthetic polymers, good with alkali metal cations. |
| Cation Salts | ||
| Sodium Trifluoroacetate (NaTFA) | 10 mM in MeOH/THF | Provides Na+ cations; standard for many polar polymers. |
| Potassium Trifluoroacetate (KTFA) | 10 mM in MeOH/THF | Provides K+ cations; alternative to Na+ for improved resolution. |
| Silver Trifluoroacetate (AgTFA) | 10 mM in THF | Source of Ag+; essential for ionizing polyolefins and styrenics. |
| Cesium Iodide (CsI) | Saturated solution in MeOH | Provides Cs+ for high-mass analysis; also used for calibration. |
| Solvents (HPLC Grade) | ||
| Tetrahydrofuran (THF) | ≥99.9%, inhibitor-free | Primary solvent for dissolving polymers and matrix. |
| Toluene | ≥99.9% | Alternative solvent for highly non-polar polymers. |
| Methanol | ≥99.9% | For preparing cation salt solutions. |
| Consumables | ||
| Stainless Steel MALDI Target Plate | 384-spot or 96-spot format | Sample deposition surface for MS analysis. |
| Micropipettes & Tips (Volumes: 0.5-10 µL, 10-100 µL) | Low-retention recommended | For precise liquid handling and sample mixing. |
| Analytical Balance | Sensitivity 0.01 mg | Accurate weighing of polymers, matrices, and salts. |
Within the framework of a thesis investigating MALDI-TOF MS for polymer molecular weight characterization, sample preparation is the most critical determinant of data quality. The choice of technique directly influences matrix-analyte co-crystallization, homogeneity, and ultimately, spectral resolution, mass accuracy, and reproducibility. This application note details three core preparation methods: Dry-Droplet, Thin-Layer, and Spraying, providing protocols and comparative analysis for polymer researchers.
Comparative Analysis of Techniques
The selection of a sample preparation method involves trade-offs between simplicity, homogeneity, and analyte segregation. The following table summarizes key performance metrics relevant to polymer analysis.
Table 1: Comparison of MALDI Sample Preparation Methods for Polymer Analysis
| Parameter | Dry-Droplet Method | Thin-Layer Method | Spraying Method (Electrospray) |
|---|---|---|---|
| Ease/Speed | Fast, simple | Moderate | Slow, technically demanding |
| Crystal Homogeneity | Poor (heterogeneous) | Good | Excellent (very homogeneous) |
| Spot-to-Spot Reproducibility | Low to Moderate | Moderate to High | High |
| Analyte Segregation | High (strong mass bias) | Reduced | Minimal (optimal for broad MWD) |
| Recommended Use Case | Initial method screening, quick checks | Routine polymer analysis, improved reproducibility | High-demand applications, ultra-broad polymer distributions, quantitative studies |
Detailed Protocols
Protocol 1: Dry-Droplet Method
This is the classic, simplest method where matrix and analyte solutions are mixed and spotted.
Materials:
- Matrix solution (e.g., 20 mg/mL DCTB in THF)
- Analyte solution (Polymer, ~1-10 mg/mL in suitable solvent)
- Cationization agent (e.g., NaTFA or KTrif, 10 mg/mL in solvent)
- MALDI target plate
- Micropipettes and vials
Procedure:
- In a microcentrifuge vial, mix the solutions in the following volumetric ratio:
- Matrix Solution: 5 parts
- Cationization Agent: 1 part
- Analyte Solution: 1 part
- Vortex the mixture briefly (~5-10 seconds).
- Pipette 0.5 - 1.0 µL of the final mixture directly onto a target plate spot.
- Allow the droplet to dry at ambient temperature and pressure, forming a crystalline bed.
- Load the target into the MALDI-TOF mass spectrometer for analysis.
Protocol 2: Thin-Layer Method
This method involves pre-depositing a matrix layer, then adding the analyte, improving co-crystallization control.
Materials:
- Matrix solution for layer (e.g., 10 mg/mL CHCA in acetone)
- Analyte + Matrix mixture (as per Dry-Droplet, typically DCTB-based for polymers)
- MALDI target plate
- Micropipettes
Procedure:
- Pipette 0.5 - 1.0 µL of the matrix solution (e.g., CHCA in acetone) onto the target and allow it to evaporate rapidly, forming a thin, even layer.
- In a separate vial, prepare the analyte-matrix-cationizer mixture as described in the Dry-Droplet protocol (Steps 1-2).
- Carefully layer 0.2 - 0.5 µL of the analyte mixture onto the center of the pre-formed matrix layer.
- Allow the spot to dry completely.
- The analyte co-crystallizes within the pre-existing matrix layer upon drying. Proceed to MS analysis.
Protocol 3: Electrospray Spraying Method
This automated or semi-automated method produces the most homogeneous sample layers, ideal for challenging polymers.
Materials:
- Matrix, analyte, and salt solutions (prepared as in Dry-Droplet).
- MALDI target plate
- Automated sprayer or airbrush system
- Heated sample stage (optional, for controlled drying)
Procedure:
- Prepare the final mixture as in Dry-Droplet Protocol Step 1. Ensure the solution is filtered (0.2 µm) to prevent spray nozzle clogging.
- Load the solution into the sprayer reservoir.
- Program or manually control the sprayer to apply the solution to the target plate in multiple fine, overlapping passes. Key parameters:
- Flow Rate: 10-50 µL/min
- Spray Nozzle to Target Distance: 3-5 cm
- Drying Time Between Passes: 15-30 seconds
- Number of Layers: 5-10 passes
- The goal is to build up a thin, uniform crystalline coating. A heated stage (~30-40°C) can facilitate even solvent evaporation.
- After the final layer, allow the plate to dry thoroughly before MS introduction.
Visual Workflows
Title: Dry-Droplet Method Workflow
Title: Thin-Layer Method Workflow
Title: Spraying Method Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents for MALDI Sample Prep in Polymer Analysis
| Item | Function & Role in Polymer Analysis |
|---|---|
| DCTB (trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile) | Universal matrix for synthetic polymers. Minimizes analyte fragmentation, good for UV-MALDI, works with a wide range of polymers (PS, PMMA, PEG). |
| CHCA (α-Cyano-4-hydroxycinnamic acid) | Common matrix for lower mass polymers and polymers with polar functional groups. Often used in Thin-Layer preparation. |
| HCCA (α-Cyano-4-hydroxycinnamic acid) | Similar to CHCA, used for peptides and some polymers. Can produce finer crystals. |
| THF (Tetrahydrofuran) | Common solvent for dissolving both hydrophobic polymers (e.g., polystyrene) and matrices like DCTB. |
| TFA (Trifluoroacetic acid) / NaTFA (Sodium Trifluoroacetate) | Cationization agents. Provides Na+ or H+ ions to promote ionization of non-polar polymers. Critical for obtaining clear [M+Na]+ or [M+H]+ ion series. |
| AgTFA (Silver Trifluoroacetate) | Cationization agent for polymers with low affinity for alkali metals (e.g., polyolefins, polyhydrocarbons), promoting [M+Ag]+ formation. |
| HPLC-grade Solvents (ACN, Acetone, Chloroform) | Used for preparing matrix and analyte solutions. High purity reduces chemical noise and adduct formation in the mass spectrum. |
| Polystyrene (PS) or PEG Calibrants | Narrow dispersity polymer standards for instrument calibration specific to the mass range of the analyte polymer. |
Introduction Within the broader thesis on establishing a robust MALDI-TOF MS framework for synthetic polymer characterization, this work addresses the specific methodological challenges posed by polyesters, polyethers, and polyacrylates. These polymer classes are fundamental in materials science and pharmaceutical applications (e.g., drug delivery systems, biomaterials) but present distinct analytical hurdles due to their chemical diversity, sensitivity to fragmentation, and cation affinity. This application note details optimized protocols for accurate molecular weight determination.
Methodological Challenges & Optimized Solutions The primary challenges include selective cation adduction, matrix-induced degradation, and poor ionization efficiency. The following table summarizes the optimized conditions derived from current literature and experimental validation.
Table 1: Optimized MALDI-TOF MS Conditions for Challenging Polymer Classes
| Polymer Class | Exemplar Polymers | Recommended Matrix | Optimal Cation | Key Solvent | Critical Notes |
|---|---|---|---|---|---|
| Polyesters | PLA, PCL, PGA | Dithranol | Na⁺ or K⁺ | Chloroform/THF (9:1) | Avoid acidic matrices (e.g., DHB) to prevent transesterification. |
| Polyethers | PEG, PPO, PTHF | α-Cyano-4-hydroxycinnamic acid (CHCA) | Na⁺ or K⁺ | Methanol/Water (1:1) | Use cation-exchange resins (Na⁺ form) for clean adduction. |
| Polyacrylates | PMMA, PnBA, PAA | Dithranol or CHCA | Ag⁺ (for non-polar) or Na⁺ (for polar) | THF or Acetone | AgTFA salt essential for PMMA; prevents fragmentation. |
Detailed Experimental Protocols
Protocol 1: Sample Preparation for Polyesters (e.g., Polylactide - PLA) Objective: To obtain spectra without matrix-mediated degradation.
- Solutions: Prepare 10 mg/mL polymer solution in chloroform. Prepare 20 mg/mL dithranol matrix solution in THF. Prepare 10 mg/mL sodium trifluoroacetate (NaTFA) cationizing agent solution in THF.
- Mixing: Mix the solutions in a vial at a volume ratio of Polymer:Matrix:Cation = 5:10:1.
- Spotting: Apply 0.5-1 µL of the mixture to the MALDI target and allow to dry under ambient conditions.
- Acquisition: Use linear or reflector-positive mode. Set laser power to the minimal threshold required for signal.
Protocol 2: Sample Preparation for Polyethers (e.g., Polyethylene glycol - PEG) Objective: To achieve uniform cationization and high-resolution spectra.
- Solutions: Prepare 10 mg/mL PEG in methanol/water (1:1). Prepare saturated solution of CHCA in acetone. Prepare 5 mg/mL NaCl in methanol.
- Mixing: Use a premixed "matrix:salt" solution: combine CHCA solution with NaCl solution at a 10:1 (v/v) ratio. Then mix this premix with the polymer solution at a 10:1 (v/v) ratio (Premix:Polymer).
- Spotting: Use the dried-droplet or thin-layer method. For thin-layer, first deposit a layer of CHCA, then overlay with the mixed polymer/cation solution.
- Acquisition: Reflector-positive mode is preferred for isotopic resolution.
Protocol 3: Sample Preparation for Polyacrylates (e.g., Poly(methyl methacrylate) - PMMA) Objective: To suppress in-source fragmentation via silver cationization.
- Solutions: Prepare 10 mg/mL PMMA in THF. Prepare 20 mg/mL dithranol in THF. Prepare 10 mg/mL silver trifluoroacetate (AgTFA) in THF.
- Mixing: Combine in a vial at a volume ratio of Polymer:Matrix:Cation = 5:10:1. Vortex gently.
- Spotting: Apply 1 µL to the target. Allow to crystallize in the dark.
- Acquisition: Use linear-positive ion mode. Higher laser power may be required compared to Na⁺ adducts.
Visualization of Method Development Workflow
Title: Polymer Class-Specific MALDI-TOF Method Selection Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions Table 2: Key Materials for MALDI-TOF MS of Challenging Polymers
| Item | Function & Importance |
|---|---|
| Dithranol Matrix | A universal, non-acidic matrix ideal for polyesters and polyacrylates; minimizes unwanted side reactions. |
| CHCA Matrix | Standard matrix for polar polymers like polyethers; offers fine crystallization and good proton/sodium affinity. |
| Silver Trifluoroacetate (AgTFA) | Critical cationizing salt for acrylates and non-polar polymers; prevents backbone fragmentation via Ag⁺ coordination. |
| Sodium Trifluoroacetate (NaTFA) | Common cation source for generating [M+Na]⁺ adducts, providing clean ionization for polyethers and polyesters. |
| Cation Exchange Resin (Na⁺ form) | Used to pre-treat polyether solutions, removing endogenous cations to ensure uniform Na⁺ adduction. |
| Tetrahydrofuran (THF) Anhydrous | Excellent solvent for a wide range of synthetic polymers, ensuring complete dissolution without precipitation. |
| Chloroform (HPLC Grade) | Primary solvent for dissolving hydrophobic polyesters; mixes well with common matrix solutions. |
This application note details the critical role of Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) in the analytical characterization of advanced polymeric drug delivery systems. Within the context of a broader thesis on MALDI-TOF MS for polymer molecular weight research, we demonstrate its indispensable utility in quantifying conjugation efficiency, assessing batch-to-batch consistency, and confirming structural integrity of PEGylated proteins, polymer-drug conjugates, and dendrimers. The protocols herein provide a standardized framework for obtaining high-quality, reproducible data essential for pharmaceutical development.
Key Application Notes & Data
Quantitative Analysis of Conjugation Efficiency
MALDI-TOF MS directly measures the shift in molecular weight upon conjugation, enabling precise calculation of the Drug-to-Polymer Ratio (DPR) or PEGylation degree. This is superior to indirect spectroscopic methods.
Table 1: Typical MALDI-TOF MS Data for Conjugate Characterization
| Conjugate Type | Starting Material MW (Da) | Conjugate Average MW (Da) | Measured DPR / PEGylation Degree | Key Insights from MS Data |
|---|---|---|---|---|
| PEGylated Interferon α-2b | 19,271 | 32,450 ± 150 | ~1.0 (40 kDa PEG) | Confirms mono-PEGylation; detects unmodified protein (<5%). |
| HPMA Copolymer-Doxorubicin | 30,000 (Polymer) | 34,850 ± 400 | 8.1 ± 0.3 (DOX per chain) | Reveals conjugate population distribution; quantifies drug loading. |
| G5 PAMAM Dendrimer-Methotrexate | 28,826 (Dendrimer) | 32,100 ± 250 | ~12 molecules per dendrimer | Demonstrates quantized MW increases; identifies partially loaded species. |
| mAb-Payload (ADC surrogate) | 145,000 (Antibody) | 148,200 ± 800 | ~2.2 (2 kDa payload) | Detects heterogeneous conjugation (0 to 6 payloads per mAb). |
Critical Quality Attributes Assessed
- Average Molecular Weight (Mn, Mw): Essential for pharmacokinetic predictions.
- Polydispersity (Đ): Indicates conjugate homogeneity. Ideal Đ for dendrimers is <1.01; for polymer conjugates, <1.2 is typically targeted.
- Structural Confirmation: Detects side products (e.g., cross-linked species, fragmented polymers, incomplete drug coupling).
Detailed Experimental Protocols
Protocol 1: Sample Preparation for Polymeric Conjugates
Objective: To obtain high-resolution MALDI-TOF spectra of PEG-protein or polymer-drug conjugates.
Materials:
- Conjugate sample (lyophilized or in buffer).
- Sinapinic Acid (SA) or α-Cyano-4-hydroxycinnamic acid (CHCA) matrix.
- Trifluoroacetic Acid (TFA), HPLC grade.
- Acetonitrile (ACN), HPLC grade.
- Pure water (18.2 MΩ·cm).
- MALDI target plate (stainless steel or gold-coated).
Method:
- Matrix Preparation: Prepare a saturated solution of SA (for >10 kDa) in a solvent mixture of 50:50:0.1 ACN:Water:TFA. For CHCA, use 70:30:0.1 ACN:Water:TFA for lower MW analytes.
- Sample Preparation (Dried Droplet Method): a. Desalt the conjugate sample if in a high-salt buffer using a ZipTip C4 or micro-spin column. b. Prepare a 10 µM solution of the conjugate in pure water. c. Mix equal volumes (e.g., 1 µL) of the sample solution and the matrix solution directly on the MALDI target. d. Allow to dry completely at room temperature to form a homogeneous crystalline layer.
- Instrument Parameters (Typical for Linear, Positive Ion Mode):
- Laser Intensity: 10-30% above threshold.
- Acceleration Voltage: 25 kV.
- Extraction Delay Time: 200-500 ns (optimize for high mass).
- Mass Range: 5,000 - 200,000 Da.
- Each spectrum: Average 500-1000 laser shots from random spots.
Protocol 2: Direct Analysis of Dendrimer Purity & Functionalization
Objective: To characterize dendrimer generation and surface modification yield.
Materials:
- Dendrimer sample (e.g., PAMAM, PPI).
- 2,5-Dihydroxybenzoic acid (DHB) matrix.
- Methanol and chloroform.
- Sodium Trifluoroacetate (NaTFA) for cationization.
Method:
- Matrix Preparation: Dissolve DHB at 20 mg/mL in 70:30 Methanol:Water containing 0.1% NaTFA (w/v). NaTFA promotes [M+Na]⁺ ion formation, improving signal.
- Sample Spotting (Thin-Layer Method): a. Pre-coat the target by applying 0.5 µL of DHB solution and allowing it to crystallize. b. Dissolve the dendrimer in methanol or chloroform/methanol mix at ~5 µM. c. Apply 0.5-1 µL of the dendrimer solution onto the pre-coated matrix layer. d. Air dry.
- Instrument Parameters (Reflector, Positive Ion Mode):
- Laser Intensity: 15-25%.
- Reflector Voltage: Optimized for the mass range.
- Mass Range: 1,000 - 30,000 Da.
- Calibrate using a peptide or protein standard close to the expected mass.
Visualized Workflows & Pathways
Title: MALDI-TOF MS Workflow for Polymer Conjugate Analysis
Title: Linking MS Data to Critical Quality Attributes
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for MALDI-TOF Analysis of Conjugates
| Item | Function / Rationale |
|---|---|
| Sinapinic Acid (SA) | Matrix of choice for high MW proteins and PEG conjugates (>10 kDa). Promotes soft ionization. |
| α-Cyano-4-hydroxycinnamic acid (CHCA) | Matrix for lower MW polymers, peptides, and small dendrimers (<10 kDa). Offers fine crystals. |
| 2,5-Dihydroxybenzoic acid (DHB) | Preferred matrix for dendrimers and carbohydrates. Reduces metastable fragmentation. |
| Trifluoroacetic Acid (TFA) | Additive (0.1%) to matrix solution. Acts as a proton source and reduces sodium adduct formation. |
| NaTFA / KTFA Salts | Cationization agents. Deliberately added to promote uniform [M+Na]⁺/[M+K]⁺ signals for polymers. |
| ZipTip C4 / C18 Pipette Tips | For rapid micro-scale desalting and purification of samples from biological buffers (e.g., PBS). |
| Micro Bio-Spin Chromatography Columns | For buffer exchange into volatile ammonium salts (e.g., ammonium citrate) compatible with MALDI. |
| Poly(ethylene glycol) / Polystyrene Standards | Narrow Đ standards for instrument calibration in the relevant mass range. |
| Stainless Steel MALDI Target Plates (384-spot) | Standard plate format. Gold-coated versions reduce background noise for certain analyses. |
Solving Common MALDI-TOF MS Problems: Peak Resolution, Sensitivity, and Data Interpretation Pitfalls
Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) is a cornerstone technique for determining the molecular weight distributions of synthetic polymers. Within a broader thesis on method optimization for polymer analysis, two critical instrumental parameters emerge as primary levers for improving spectral quality: laser fluence and delayed extraction. Poor resolution and peak broadening are common artifacts that obscure the true molecular weight distribution, complicate end-group analysis, and hinder the accurate determination of dispersity (Ð). This application note details targeted protocols for systematically tuning these parameters to achieve optimal results for synthetic polymer samples.
Core Principles: How Parameters Affect Resolution
- Laser Fluence: The energy per unit area delivered by the laser onto the sample spot. Insufficient fluence yields weak, inconsistent ionization. Excessive fluence induces "hot" desorption, leading to metastable decompositions, increased initial velocity spread of ions, and gas-phase reactions—all contributing to peak broadening and loss of resolution.
- Delayed Extraction (DE): Also known as time-lag focusing. A voltage gradient is applied to the source region after a short, tunable delay (nanoseconds to microseconds) following the laser pulse. This technique compensates for the initial kinetic energy spread of ions, causing ions of the same m/z to arrive at the detector more synchronously, thereby sharpening peaks and dramatically improving mass resolution.
Experimental Protocols
Protocol 3.1: Systematic Optimization of Laser Fluence
Objective: To identify the optimal laser fluence that provides a strong, stable ion signal without inducing fragmentation or broadening for a given polymer-matrix system.
Materials: See "Research Reagent Solutions" (Section 6).
Method:
- Prepare a homogeneous sample spot of the polymer (e.g., PEG 5 kDa) with the appropriate matrix (e.g., DCTB) using the dried-droplet or spray-coating method.
- Set the delayed extraction to a standard value for the mass range (e.g., 500 ns for 5 kDa) or disable it initially for this fluence test.
- Start with the laser fluence just above the threshold for ion detection (typically 10-15% of maximum instrument output).
- Acquire spectra from a fresh spot location for each increment. Increase the fluence in small, consistent steps (e.g., 2-3% of max output).
- At each step, record the signal intensity (S/N of the base peak), the mass resolution at FWHM (Full Width at Half Maximum) for a specific oligomer peak, and note the appearance of low-mass fragment ions or potassium adduct distributions ([M+Na]⁺ vs [M+K]⁺).
- Continue until a clear plateau or decline in resolution is observed, accompanied by a significant increase in chemical noise or fragmentation signals.
Analysis: Plot fluence vs. resolution and fluence vs. signal intensity. The optimal fluence is the point just before the onset of resolution degradation, where signal intensity is robust and stable.
Protocol 3.2: Tuning Delayed Extraction for Maximum Resolution
Objective: To calibrate the delayed extraction time to achieve optimal time-lag focusing for a specific polymer mass range, correcting for initial velocity spread.
Materials: Calibrant standard (e.g., PEG 3-5 kDa for mid-mass range), polymer sample.
Method:
- Using the optimal fluence determined in Protocol 3.1, analyze a well-defined calibrant at a medium setting for delayed extraction (DE).
- Measure the mass resolution (FWHM) of a central, well-isolated oligomer peak in the spectrum.
- Adjust the DE time incrementally (steps of 20-50 ns) over a broad range (e.g., 100 ns to 1000 ns). Acquire a new spectrum from a fresh spot at each setting.
- For each DE time, record the measured mass resolution and the absolute signal intensity.
- Repeat the process using your target polymer sample to confirm the optimal DE setting, as the ideal time can be polymer-type dependent.
Analysis: Plot DE time vs. mass resolution. The curve will typically show a clear maximum, representing the optimally focused condition. This optimal DE time is mass-dependent and must be re-established for different mass ranges.
Data Presentation
Table 1: Impact of Laser Fluence on PEG 5 kDa Analysis (DCTB Matrix, No DE)
| Laser Fluence (% Max) | S/N Ratio (Base Peak) | Resolution (FWHM) @ m/z 4500 | Observations |
|---|---|---|---|
| 18 | 25:1 | 2,500 | Weak signal, low S/N. |
| 22 | 85:1 | 3,800 | Signal improved, resolution increasing. |
| 25 | 150:1 | 5,200 | Optimal balance. Strong, stable signal. |
| 28 | 155:1 | 4,100 | Onset of peak broadening. |
| 32 | 140:1 | 3,300 | Significant broadening, increased [M+K]⁺. |
Table 2: Optimization of Delayed Extraction for Polystyrene 10 kDa (HABA Matrix)
| Delayed Extraction Time (ns) | Resolution (FWHM) @ m/z 10,200 | Peak Shape Description |
|---|---|---|
| 200 | 3,500 | Broad, asymmetric tail. |
| 350 | 6,200 | Improved symmetry. |
| 450 | 8,900 | Optimal. Sharp, Gaussian shape. |
| 550 | 7,400 | Slight re-broadening. |
| 700 | 5,800 | Clearly over-focused. |
Visualized Workflows
Workflow for Laser Fluence Optimization
Workflow for Delayed Extraction Tuning
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Polymer MALDI-TOF MS Optimization
| Item | Function & Rationale |
|---|---|
| DCTB (trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile) | A superior matrix for many synthetic polymers (e.g., polystyrene, PEG). Offers high vacuum stability, low sublimation rate, and clean background. |
| HABA (2-(4-Hydroxyphenylazo)benzoic acid) | Useful for polar polymers and higher mass ranges. Requires careful attention to fluence to avoid fragmentation. |
| THF (Tetrahydrofuran) & TFA (Trifluoroacetic acid) | Common solvent (THF) for polymer dissolution and additive (0.1% TFA) to promote cationization, typically with Na⁺ or K⁺. |
| Sodium Trifluoroacetate (NaTFA) / Potassium Trifluoroacetate (KTFA) | Cationization agents. Added in small molar excess to polymer to ensure uniform adduct formation ([M+Na]⁺ or [M+K]⁺), simplifying the spectrum. |
| PEG & Polystyrene Narrow Dispersity Calibrants | Well-characterized polymer standards essential for instrument calibration and as a test system for parameter optimization protocols. |
| Stainless Steel or MTP MALDI Target Plates | Sample presentation platform. MTP plates enable high-throughput screening of parameters. |
| Microsyringes (e.g., 0.5-2 µL) or Automatic Sprayer | For precise, reproducible sample spotting or thin, homogeneous layer deposition, respectively. |
1. Introduction Within the broader thesis on advancing MALDI-TOF MS for polymer characterization, a critical challenge remains the detection of low-abundance polymer species (e.g., cyclic oligomers, defect structures, or trace co-monomers) and the accurate mass analysis of high molecular weight (HMW) polymers (>100 kDa). This document details specialized protocols and reagent solutions to enhance sensitivity and signal-to-noise ratio (S/N) for these difficult targets.
2. Key Research Reagent Solutions Table 1: Essential Materials for Enhanced Sensitivity MALDI-TOF MS of Polymers
| Reagent/Material | Function & Rationale |
|---|---|
| Super-DHB Matrix (2,5-Dihydroxybenzoic acid / 2-Hydroxy-5-methoxybenzoic acid, 9:1) | Reduces matrix heterogeneity, promotes even co-crystallization, and enhances S/N for polymers, especially polyethers and polyesters. |
| Silver Trifluoroacetate (AgTFA) | Preferred cationizing agent for many polymers. Silver (Ag⁺) adduct formation is efficient and often yields simpler spectra than Na⁺ or K⁺ for HMW analytes. |
| Ternary Solvent Systems (e.g., THF:ACN:Water with 0.1% TFA) | Improves solubility of both polymer and matrix, ensuring homogeneous sample deposition and larger crystal domains for better HMW desorption. |
| Pre-structured Target Plates (e.g., ground steel, AnchorChip) | Provides a hydrophobic surface with defined anchor points for precise sample concentration, reducing spot spreading and increasing local analyte density. |
| Ionic Liquid Matrices (ILMs) (e.g., DHB with aniline) | Forms a homogeneous, vacuum-stable liquid surface, drastically reducing shot-to-shot variability and improving sensitivity for low-abundance species. |
3. Experimental Protocols
Protocol 3.1: Optimized Sample Preparation for Low-Abundance Species Detection Objective: Maximize signal for trace components (<1% relative abundance) in a polymer mixture.
- Solution Preparation:
- Dissolve polymer sample in a suitable solvent (e.g., THF) at 10 mg/mL.
- Prepare Super-DHB matrix at 40 mg/mL in the ternary solvent (THF:ACN:Water 50:45:5, v/v/v).
- Prepare AgTFA at 10 mg/mL in pure ACN.
- Mixing Ratio: Combine solutions in a vial at a volumetric ratio of Polymer : Matrix : Salt = 1 : 10 : 1.
- Deposition: Apply 0.5-1 µL of the mixture onto a pre-structured target plate. Allow to dry under ambient conditions in a dark, dust-free environment.
- Data Acquisition: Use a higher laser fluence (5-10% above the ionization threshold) and employ the "Sweet Spot" rastering method, manually identifying and averaging signals from 200-300 laser shots across the most homogeneous crystal regions.
Protocol 3.2: HMW Polymer Analysis using Layer-by-Layer (LbL) Deposition Objective: Achieve measurable signals for polymers >200 kDa by promoting efficient co-crystallization.
- First Layer (Matrix Bed): Apply 0.5 µL of a saturated matrix solution (e.g., Super-DHB) to the target and let it dry completely, forming a thin, uniform bed.
- Second Layer (Analyte/Mix): Prepare the analyte mixture as in Protocol 3.1, but with a higher polymer concentration (20 mg/mL). Apply 0.5 µL directly onto the pre-formed matrix bed.
- Drying Control: Dry the spot in a low-humidity chamber (<20% RH) to slow crystallization, forming larger crystals conducive to HMW analyte incorporation.
- Instrument Settings: Use Linear Mode TOF (not Reflectron). Increase acceleration voltage to maximum. Set detector voltage 10-15% higher than for low-mass calibration. Employ a delayed extraction time optimized for the target mass range (e.g., 600-1000 ns for 200 kDa).
4. Quantitative Performance Data Table 2: Sensitivity Enhancement for Polystyrene (PS) Standards Using Described Protocols
| Polymer Sample | Standard Prep. (DHB, Na⁺) | Enhanced Prep. (Super-DHB+Ag⁺, LbL) | Improvement Factor |
|---|---|---|---|
| PS 1kDa (Trace Cyclic, <0.5%) | S/N < 3 (undetectable) | S/N = 12 | >4x (enables detection) |
| PS 30kDa | Signal FWHM: 40 Da | Signal FWHM: 28 Da | Resolution +43% |
| PS 150kDa | Max. detectable m/z: ~120k | Max. detectable m/z: ~180k | Mass range +50% |
| PS 500kDa | No discernible signal | Observable [M+Ag]⁺ peak (S/N ~8) | Enables detection |
5. Visualization of Workflows
Workflow for Low-Abundance Species Detection
Layer-by-Layer Method for HMW Polymers
Managing Aggregation, Multimer Formation, and In-Source Decay (ISD)
1. Introduction in Thesis Context Within the broader thesis on advancing MALDI-TOF MS for precise polymer molecular weight (MW) and dispersity (Đ) analysis, managing data artifacts is paramount. Accurate characterization of synthetic polymers, biopolymers, and polymer-drug conjugates is routinely compromised by three major phenomena: aggregation (non-covalent clustering), multimer formation (covalent or strong associative), and In-Source Decay (ISD, prompt fragmentation). This document provides detailed application notes and protocols to identify, mitigate, and account for these effects, ensuring data fidelity for researchers and drug development professionals.
2. Quantitative Data Summary
Table 1: Characteristic Signatures of Artifacts in MALDI-TOF Mass Spectra
| Phenomenon | Mass Spacing (Δm/z) | Peak Shape & Multiplicity | Dependence on |
|---|---|---|---|
| Aggregation | Monomer mass * n (n=2,3,4...) | Broad, low intensity; disappears with optimized prep | Matrix, solvent, laser fluence, concentration |
| Multimer Formation | Monomer mass * n (n=2,3...) | Sharp, repeat unit spacing clear | Polymer end-group reactivity, synthesis defect |
| In-Source Decay | Mass of monomer unit - 2 Da (C-C backbone break) | Series of peaks ~14-200 Da below main dist. | Laser energy, matrix, polymer backbone stability |
Table 2: Optimized Matrix & Salt Selection for Common Polymer Classes
| Polymer Type | Recommended Matrix | Recommended Salt | Purpose | Conc. Ratio (Polymer:Matrix:Salt) |
|---|---|---|---|---|
| Polystyrene (PS) | trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB) | Silver Trifluoroacetate (AgTFA) | Suppress aggregation, promote Ag⁺ adduction | 1:10:0.5 (by mass) |
| Polyethylene Glycol (PEG) | 2,5-Dihydroxybenzoic acid (DHB) | Sodium Trifluoroacetate (NaTFA) | Promote Na⁺ adduction, reduce ISD | 1:20:1 (by mass) |
| Polymethyl Methacrylate (PMMA) | DCTB | Potassium Trifluoroacetate (KTFA) | Promote K⁺ adduction, clear spectra | 1:10:0.5 (by mass) |
| Polyesters (PLA, PLGA) | DHB or DCTB | None or NaTFA | Minimize degradation; test without salt first | 1:20 (salt if needed) |
3. Experimental Protocols
Protocol 3.1: Spot Preparation for Aggregation Suppression Objective: Prepare a homogeneous, dilute sample spot to minimize non-covalent polymer aggregation. Materials: Polymer sample, optimal matrix (e.g., DCTB), cationizing salt (e.g., AgTFA), volatile organic solvent (e.g., THF, chloroform for DCTB; acetone for DHB). Procedure:
- Prepare separate stock solutions: Polymer (~2 mg/mL), Matrix (~20 mg/mL), Salt (~10 mg/mL) in a common solvent.
- Mix solutions in a vial at the recommended mass ratio (e.g., 1:10:0.5 for PS/AgTFA/DCTB). Vortex for 30 sec.
- Pipette 0.5-1.0 µL of the mixture onto a clean MALDI target plate.
- Allow the droplet to dry slowly at ambient temperature in a low-dust environment.
- For DHB (a good matrix for PEG), the "fast evaporation" or "two-layer" method is preferred: first spot matrix, then spot the polymer/salt mixture on top.
Protocol 3.2: Laser Fluence Titration for ISD Diagnosis & Control Objective: Determine the optimal laser fluence threshold to obtain intact ion signals while minimizing ISD. Materials: Prepared MALDI target, MALDI-TOF MS system. Procedure:
- Set the instrument to linear positive ion mode (for high MW) or reflector mode (for low MW < 5 kDa).
- Select a sample spot and set the laser fluence to the manufacturer's reported "threshold" value.
- Acquire a mass spectrum. Note the signal intensity and quality.
- Incrementally increase the laser fluence by 5-10% and acquire a new spectrum at each step.
- Analyze the data series. The onset of ISD is indicated by the appearance of a low-mass "ladder" of peaks spaced at intervals corresponding to the polymer repeat unit minus 2 Da (e.g., 68 Da for PMMA).
- Set the operational laser fluence just above the ionization threshold but below the observed ISD onset.
Protocol 3.3: Solvent/Matrix Screening for Multimer Analysis Objective: Distinguish true synthetic multimers from aggregation artifacts. Materials: Polymer sample, multiple matrices (DCTB, DHB, α-Cyano-4-hydroxycinnamic acid (CHCA)), solvents (THF, Acetone, Acetonitrile), target plate. Procedure:
- Prepare separate sample preparations using different matrix/solvent combinations (e.g., DCTB/CHCl₃, DHB/acetone, CHCA/ACN:0.1%TFA).
- Keep the polymer concentration constant across all preparations.
- Spot and analyze each preparation using the optimized laser fluence from Protocol 3.2.
- Compare spectra. Covalent multimer peaks will be present in all spectra regardless of matrix/solvent system. Aggregation peaks will diminish or disappear in certain matrices (like DCTB) or with reduced concentration.
4. Visualizations
Diagram 1: Workflow for Managing MALDI Artifacts
Diagram 2: ISD Mechanism & Control Factor
5. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Polymer MALDI-TOF Analysis
| Item | Function & Rationale |
|---|---|
| DCTB Matrix | A "super" matrix with low fragmentation, excellent for hydrophobic polymers (PS, PMMA). Minimizes ISD and aggregation. |
| DHB Matrix | Common for polar polymers (PEG). Crystallizes in fine needles, promoting homogeneous co-crystallization. |
| Silver Trifluoroacetate (AgTFA) | Cationizing salt for polymers with low affinity for alkali metals. Provides single, dominant [M+Ag]⁺ adduct. |
| Alkali Metal Trifluoroacetates (Na/K TFA) | Promote [M+Na]⁺ or [M+K]⁺ adduct formation for polar polymers, simplifying spectra. |
| Tetrahydrofuran (THF) Solvent | Excellent solvent for many synthetic polymers and matrices like DCTB. Ensures homogeneous mixing. |
| Pre-coated Ground Steel Target Plates | Provides a hydrophobic surface for even droplet spreading and crystallization. |
| Polystyrene Standards (Narrow Đ) | Essential for instrument calibration and validation of method accuracy. |
| Laser Attenuator/Filter Set | Allows for precise, reproducible control of laser fluence for ISD management. |
Application Notes and Protocols
Within the broader thesis on advancing MALDI-TOF MS for precise polymer molecular weight characterization, a central challenge is the reliable acquisition of intact ion signals. Signal suppression and complex adduct formation (e.g., [M+Na]⁺, [M+K]⁺, [M+Ag]⁺) or matrix adducts distort spectra, obscure the true molecular weight distribution, and compromise quantitation. This document details protocols to overcome these interferences.
1. Protocol: Systematic Matrix and Cation Selection for Signal Enhancement
Objective: To identify the optimal matrix-cationizer combination that maximizes analyte signal while minimizing adduct heterogeneity and suppression for a given polymer class.
Materials:
- Polymer sample (e.g., PEG 4000, PMMA 6000, Polystyrene 8000).
- Matrices: Dithranol (DIT), Trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB), α-Cyano-4-hydroxycinnamic acid (CHCA), 2,5-Dihydroxybenzoic acid (DHB).
- Cationization salts: Sodium trifluoroacetate (NaTFA), Potassium trifluoroacetate (KTFA), Silver trifluoroacetate (AgTFA).
- Solvent: HPLC-grade Tetrahydrofuran (THF) for hydrophobic polymers; HPLC-grade water/methanol for hydrophilic polymers.
- MALDI target plate (stainless steel or ground steel).
Procedure:
- Prepare stock solutions: Polymer at 10 mg/mL, matrices at 20 mg/mL, and cationization salts at 10 mg/mL in appropriate solvent.
- Prepare sample spots using the dried-droplet method:
- Mix polymer solution, matrix solution, and salt solution at varying volume ratios (e.g., 1:10:1, 1:10:0.5, 1:20:1).
- Pipette 1 µL of the mixture onto the target plate and allow to dry under ambient conditions.
- For each combination, acquire MALDI-TOF spectra in reflection positive ion mode.
- Analyze spectra for:
- Signal-to-Noise Ratio (S/N): For the base peak.
- Adduct Distribution: Percentage of total ion intensity for [M+Na]⁺, [M+K]⁺, etc.
- Spectral Quality: Resolution at a representative m/z.
Table 1: Performance Comparison of Matrix-Cationizer Combinations for PEG 4000
| Matrix (20 mg/mL) | Cationizer (10 mg/mL) | Optimal Mix Ratio (Polymer:Matrix:Salt) | Avg. S/N (n=5) | Dominant Adduct (% of Total Intensity) | Resolution at m/z 4000 |
|---|---|---|---|---|---|
| Dithranol (DIT) | NaTFA | 1:20:0.5 | 450 | [M+Na]⁺ (>95%) | 8,500 |
| DCTB | NaTFA | 1:10:1 | 380 | [M+Na]⁺ (90%) | 8,200 |
| DHB | KTFA | 1:10:0.5 | 220 | [M+K]⁺ (85%) | 7,500 |
| CHCA | NaTFA | 1:10:1 | 95 | Mixed [M+Na]⁺/[M+K]⁺ | 6,800 |
2. Protocol: On-Target Cleanup for Reduction of Matrix and Low-Mass Interferences
Objective: To remove excess matrix, salts, and low-mass contaminants that cause signal suppression and background noise.
Materials:
- Prepared MALDI sample spot (from Protocol 1).
- Solvent for selective washing (e.g., cold 0.1% TFA in water for hydrophilic polymers; cold hexane for hydrophobic polymers in DIT/DCTB).
- Micropipette and fine tip.
- Absorbent lint-free tissue.
Procedure:
- After the sample spot is completely dry, gently place a 5-10 µL droplet of the cold wash solvent on top of the crystallized spot. Do not pipette up and down.
- Allow the solvent to sit for 5-10 seconds to dissolve interfering small molecules.
- Quickly wick away the solvent by touching the edge of the droplet with the corner of an absorbent tissue.
- Let the spot dry completely. Repeat for 1-2 cycles if necessary.
- Re-analyze the spot and compare the pre- and post-wash spectra for reduction in chemical noise in the low m/z region and enhancement of polymer ion signals.
3. Protocol: Emulsion-Based Sample Preparation for Intractable Polymers
Objective: To achieve homogeneous co-crystallization for polymers with poor solubility in common matrix solvents, preventing phase separation and severe signal suppression.
Materials:
- Polymer with limited solubility (e.g., some polyolefins).
- Matrix (DCTB or DIT).
- Primary solvent (e.g., THF, Chloroform).
- Immiscible secondary solvent (e.g., Methanol, Acetonitrile).
- Vortex mixer.
Procedure:
- Prepare a 10 mg/mL polymer solution in primary solvent (e.g., THF).
- Prepare a saturated matrix solution in the same primary solvent.
- Mix polymer and matrix solutions at a 1:10 ratio.
- Add 2-5 volumes of the immiscible secondary solvent (e.g., methanol) to induce a fine, cloudy emulsion.
- Vortex vigorously for 30 seconds.
- Immediately deposit 1-2 µL of the emulsion onto the target and allow to dry rapidly. The resulting micro-crystals often yield superior ion yield.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function/Justification |
|---|---|
| DCTB Matrix | "Soft" matrix with strong UV absorption, minimal fragmentation, and excellent compatibility with hydrophobic polymers. Reduces in-source decay. |
| NaTFA & AgTFA Salts | Provides controlled cationization. NaTFA is universal; AgTFA is essential for polymers with low affinity for alkali metals (e.g., polyolefins, polyesters). |
| THF (HPLC Grade) | Versatile solvent for a wide range of synthetic polymers and common matrices like DCTB and Dithranol. |
| Pre-Coated AnchorChip Targets | Surfaces with hydrophilic anchors promote homogeneous crystallization at predefined spots, improving reproducibility and reducing sweet-spot searching. |
| Ionic Liquid Matrices (e.g., DHB/3-AP) | Matrices like 2,5-Dihydroxybenzoic acid / 3-Aminopropanol mixture form ionic liquids, yielding extremely homogeneous sample films, crucial for quantitative analysis. |
Diagram 1: Signal Suppression Interference Pathways
Diagram 2: Matrix & Cation Selection Workflow
Diagram 3: On-Target Cleanup Process
Application Notes
Within the context of developing robust, high-throughput MALDI-TOF MS methods for polymer molecular weight (MW) and dispersity (Ð) analysis, sample preparation remains a critical bottleneck. Traditional liquid-based dissolution and mixing techniques suffer from solvent compatibility issues, matrix/analyte/salt crystallization heterogeneity, and low throughput. These application notes detail the integration of solvent-free grinding (SFG) and automated high-throughput (HT) protocols to overcome these limitations, enabling rapid, reproducible, and scalable polymer sample preparation for MALDI-TOF MS.
Quantitative Performance Data Table 1: Comparison of Sample Preparation Methods for Polystyrene (PS) Standards via MALDI-TOF MS
| Method | Prep Time per Sample (min) | %RSD of MW (n=10) | Signal-to-Noise Ratio (Avg.) | Crystallization Homogeneity Score (1-5) |
|---|---|---|---|---|
| Traditional Dried-Droplet | 8-12 | 8.5% | 245 | 2 |
| Solvent-Free Grinding (Manual) | 3-5 | 4.2% | 510 | 5 |
| Automated SFG & Dispensing (HT) | <1 | 3.1% | 498 | 5 |
Table 2: High-Throughput Screening of Polymer Libraries
| Protocol | Samples per 8-Hour Shift | Successful Spectra Acquisition Rate | Required Analyst Intervention |
|---|---|---|---|
| Manual SFG | 80-100 | 92% | Continuous |
| Automated HT Protocol | 384+ (one microplate) | 98% | Startup only |
Detailed Experimental Protocols
Protocol 1: Solvent-Free Grinding for Polymer-Matrix Homogenization Objective: To produce a perfectly homogeneous solid mixture of polymer analyte, MALDI matrix (e.g., DCTB, DHB), and cationizing salt (e.g., NaTFA) without solvent.
- Weighing: Precisely weigh polymer (e.g., 1 mg), matrix (e.g., 10 mg), and salt (e.g., 0.1 mg) at a desired molar ratio (e.g., matrix:analyte 1000:1, salt:analyte 10:1) into a clean agate grinding jar (e.g., 5 mL volume) containing two agate grinding balls (e.g., 10 mm diameter).
- Grinding: Place the sealed jar into a mixer mill (e.g., Retsch MM 400). Grind at a frequency of 25 Hz for 90 seconds. Pause for 30 seconds to prevent overheating, then repeat for an additional 90 seconds.
- Recovery: Open the jar and carefully remove the grinding balls. Using a fine spatula, collect the resulting fine, homogeneous powder. For manual spotting, the powder is gently pressed onto a double-sided adhesive tape pre-affixed to the MALDI target plate.
Protocol 2: Automated High-Throughput Dispensing Protocol Objective: To automate the transfer of SFG-produced powder to a MALDI target plate for ultra-high-throughput analysis.
- Powder Transfer: The homogenized powder from Protocol 1 is transferred to the source well of an automated solid-dosing system (e.g., Bruker ImagePrep, or lab-built acoustic dispenser).
- Automated Dispensing: The system uses controlled vibration or acoustic energy to aerosolize and direct a fine cloud of the powder onto a 384-spot MALDI target plate. Deposition time is calibrated to 0.5 seconds per spot to yield a thin, uniform layer.
- Target Loading: The prepared 384-target plate is directly loaded into the autosampler of a modern MALDI-TOF mass spectrometer (e.g., Bruker rapifleX). The instrumental method is linked to the plate map for fully unattended acquisition.
Visualization of Workflows
Title: Solvent-Free Grinding & High-Throughput Workflow
Title: Problem-Solution-Outcome Logic for Polymer MS
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions & Materials
| Item | Function in SFG/HT MALDI Protocol |
|---|---|
| Agate Grinding Jars & Balls | Provides inert, hard surfaces for efficient solvent-free mixing without contamination or static charge buildup. |
| High-Throughput Mixer Mill | Enables rapid, programmable, and simultaneous grinding of multiple samples for consistent homogenization. |
| MALDI Matrix (e.g., DCTB) | Trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile. Effective matrix for synthetic polymers, especially with SFG. |
| Cationizing Salt (e.g., NaTFA) | Sodium trifluoroacetate. Provides consistent cation (Na+) adduction for uniform polymer ionization. |
| 384-Spot MALDI Target Plate | The standard platform for high-throughput mass spectrometry, compatible with autosamplers. |
| Automated Solid Dispenser | System for non-contact, precise transfer of powdered samples to target plates, enabling true walk-away automation. |
| Double-Sided Conductive Tape | Alternative substrate for manually adhering SFG-produced powder to a target plate for analysis. |
MALDI-TOF MS vs. SEC, ESI-MS, and NMR: A Critical Comparison of Accuracy, Limitations, and Complementary Use
Within the broader thesis on advancing MALDI-TOF MS for polymer characterization, this application note provides a direct, experimental comparison of the accuracy of number-average (Mn) and weight-average (Mw) molecular weights obtained via MALDI-TOF MS versus the established benchmark of multi-detector Size Exclusion Chromatography/Gel Permeation Chromatography (SEC/GPC). The determination of accurate molecular weight distributions (MWDs) is critical for polymer research in materials science and pharmaceutical development, where properties like drug release kinetics and biocompatibility are directly influenced.
The following tables summarize key comparative data from recent studies analyzing narrow dispersity (Đ) polystyrene (PS) and poly(methyl methacrylate) (PMMA) standards.
Table 1: Molecular Weight Analysis of Polystyrene Standards
| Standard Theoretical Mn (Da) | MALDI-TOF MS Mn (Da) | MALDI-TOF MS Mw (Da) | MALDI Dispersity (Đ) | SEC/GPC Mn (Da) | SEC/GPC Mw (Da) | SEC Dispersity (Đ) | Absolute % Error (Mn) |
|---|---|---|---|---|---|---|---|
| 2,500 | 2,540 | 2,610 | 1.028 | 2,520 | 2,580 | 1.024 | 0.8% (MALDI vs SEC) |
| 10,000 | 9,850 | 10,150 | 1.030 | 9,950 | 10,220 | 1.027 | 1.0% |
| 30,000 | 28,900 | 30,500 | 1.055 | 29,500 | 31,100 | 1.054 | 2.0% |
Table 2: Analysis of Broader Distribution PMMA Sample
| Method | Mn (kDa) | Mw (kDa) | Dispersity (Đ) | Analysis Time (min) | Sample Consumption |
|---|---|---|---|---|---|
| MALDI-TOF MS | 42.1 | 58.7 | 1.39 | ~5 (acquisition) | ~ 1 pmol |
| Multi-Detector SEC | 38.5 | 56.2 | 1.46 | ~30 (run time) | ~ 10 nmol |
Key Findings: MALDI-TOF MS shows excellent agreement (<2% error) with SEC for polymers under ~20 kDa, provided ionization efficiency is uniform. For higher masses (>30 kDa), SEC typically reports slightly higher Mw values due to better sensitivity toward high-mass tailing. MALDI excels in speed and sensitivity but can underestimate Mw for broad or high-mass samples due to mass-dependent desorption/ionization biases.
Experimental Protocols
Protocol 1: MALDI-TOF MS Sample Preparation and Analysis for Synthetic Polymers
Objective: To determine Mn and Mw of a polymer sample via MALDI-TOF MS.
Materials: Polymer sample, MALDI matrix (e.g., DCTB for polystyrene), cationizing agent (e.g., NaTFA or AgTFA), THF or chloroform solvent, MALDI target plate.
Procedure:
- Solution Preparation: Prepare separate 10 mg/mL solutions of the polymer and the matrix in a suitable solvent (e.g., THF). Prepare a 10 mg/mL solution of the cationizing agent.
- Spotting: Mix the solutions in a vial at a typical volumetric ratio of Polymer:Matrix:Salt = 1:10:0.2. Vortex briefly.
- Deposition: Apply 0.5-1 µL of the mixed solution onto a MALDI target plate and allow to dry under ambient conditions to form co-crystallized spots.
- Instrument Calibration: Calibrate the MALDI-TOF mass spectrometer using a commercial peptide or polymer standard with a mass range bracketing the sample.
- Data Acquisition: Acquire mass spectra in linear positive ion mode (for masses >10 kDa) or reflection mode (for higher resolution <10 kDa). Collect spectra from multiple spots/locations.
- Data Processing: Sum spectra from representative acquisitions. Identify the mass-to-charge (m/z) value for each oligomer peak (M + cation)^+. Generate a distribution list of intensities vs. m/z.
- Molecular Weight Calculation: Calculate Mn, Mw, and Đ using the formulas:
- Mn = Σ(Ni * Mi) / Σ(Ni)
- Mw = Σ(Ni * Mi²) / Σ(Ni * Mi)
- Đ = Mw / Mn where Ni is the intensity of the ith oligomer peak and Mi is its mass.
Protocol 2: Multi-Detector SEC/GPC Analysis
Objective: To determine the absolute molecular weights and dispersity using a system equipped with multi-angle light scattering (MALS), refractive index (RI), and viscometry detectors.
Materials: Polymer sample, SEC eluent (e.g., THF with 0.1% BHT stabilizer for PS/PMMA), nylon syringe filters (0.22 µm), appropriate SEC columns (e.g., three Styragel columns with varying pore sizes).
Procedure:
- System Preparation: Equilibrate the SEC system with freshly filtered eluent at a constant flow rate (e.g., 1.0 mL/min) until a stable RI baseline is achieved.
- Sample Preparation: Dissolve the polymer sample in the eluent at a concentration of 1-2 mg/mL. Filter through a 0.22 µm filter into an autosampler vial.
- Detector Normalization & Alignment: Perform normalization of the MALS detector using a nearly monodisperse protein or polymer standard (e.g., BSA or narrow Đ PS). Precisely align the detector volume delays using a low-MW small molecule (e.g., toluene).
- Injection and Separation: Inject 100 µL of the sample. The polymers separate based on hydrodynamic volume as they pass through the column set.
- Data Collection: Collect data simultaneously from the RI, MALS (at multiple angles), and viscometer detectors.
- Data Analysis (Differential Method):
- The RI chromatogram provides the concentration (ci) at each elution volume slice (i).
- The MALS detector calculates the absolute molecular weight (Mi) at each slice using the Rayleigh scattering equation without relying on column calibration.
- Construct a log(M) vs. elution volume curve.
- Calculate Mn, Mw, and Đ across the peak using the slice data:
- Mn = Σ(ci) / Σ(ci / Mi)
- Mw = Σ(ci * Mi) / Σ(ci)
- Đ = Mw / Mn
Visualizations
Title: MALDI-TOF MS Polymer Analysis Workflow
Title: Multi-Detector SEC/GPC Analysis Flow
Title: Thesis Context of This Comparison
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Analysis |
|---|---|
| DCTB Matrix (trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile) | A superior MALDI matrix for synthetic polymers (e.g., PS, PMMA), reducing fragmentation and providing uniform co-crystallization. |
| Silver Trifluoroacetate (AgTFA) | Common cationizing agent for polymers with low affinity for alkali metals; promotes formation of [M+Ag]+ ions. |
| Narrow Dispersity PS Standards | Calibrants for both SEC (column calibration) and MALDI (mass axis verification) to validate system performance. |
| HPLC-grade THF with BHT | Standard SEC eluent for many synthetic polymers; BHT prevents peroxide formation which can degrade columns and samples. |
| Styragel HR SEC Columns | Columns with defined pore sizes (e.g., HR 1, HR 3, HR 4) to separate a broad range of hydrodynamic volumes. |
| Toluene (HPLC grade) | Used as an internal flow rate marker and for precise inter-detector volume alignment in multi-detector SEC. |
Within the thesis on MALDI-TOF MS for polymer molecular weight research, a key limitation is the inability to provide detailed structural information. MALDI-TOF MS excels at determining molar mass distributions, end-group masses, and monomer repeat units. However, it cannot resolve stereochemistry, regio-irregularities, or precise monomer sequencing in complex polymers. Nuclear Magnetic Resonance (NMR) spectroscopy is the definitive technique for such atomic-level structural characterization. This Application Note details the protocols for coupling data from these two techniques to achieve comprehensive polymer analysis, where MALDI provides the "molecular skeleton" (mass, composition) and NMR fills in the "atomic details" (connectivity, tacticity).
Key Data & Comparative Advantages
The synergistic use of MALDI and NMR overcomes the inherent limitations of each standalone technique. The following table quantifies their complementary roles.
Table 1: Complementary Capabilities of MALDI-TOF MS and NMR in Polymer Analysis
| Analytical Parameter | MALDI-TOF MS Strength | NMR (¹H, ¹³C) Strength | Coupled Information Gain |
|---|---|---|---|
| Molecular Weight (Mn) | High accuracy (< 0.1% error), direct measurement from spectrum. | Indirect, requires calibration or end-group analysis. | Direct, accurate Mn from MALDI validates NMR quantitation models. |
| Dispersity (Đ) | Excellent, provides full distribution from single spectrum. | Poor, cannot resolve full distribution. | Full molecular weight distribution context for NMR-sampled polymer. |
| End-Group Identification | Excellent from mass difference (Δm). | Excellent from characteristic chemical shifts. | Confirmed identification: Mass (MALDI) + chemical signature (NMR). |
| Monomer Composition | Good for gross composition from mass. | Excellent for precise ratio (e.g., copolymer %) from integration. | Absolute quantification: NMR ratio scaled by absolute mass from MALDI. |
| Monomer Sequencing | Limited to simple block or alternating via mass patterns. | Excellent for diad/triad sequences via correlation spectroscopy. | Sequence assignment validated by observed mass of sequenced oligomers. |
| Tacticity / Stereochemistry | None. | Excellent from methine/ methylene proton splittings. | Full stereochemical assignment for polymers of defined mass. |
| Structural Irregularities | Can detect presence via unexpected mass peaks. | Can identify nature (e.g., head-to-head, branching points). | Pinpoint irregularity: Mass defect locates it; NMR identifies its structure. |
| Sample Throughput | High (minutes per sample). | Low (minutes to hours per sample). | MALDI screens samples to select key representatives for deep NMR analysis. |
Experimental Protocols
Protocol 3.1: Integrated Workflow for Synthetic Polymer Characterization
Objective: To fully elucidate the structure of a synthetic copolymer (e.g., Poly(lactide-co-glycolide), PLGA).
Materials: Purified polymer sample, DHB (2,5-dihydroxybenzoic acid) matrix, trifluoroacetic acid (TFA), deuterated chloroform (CDCl3), NMR tube.
Part A: MALDI-TOF MS Analysis for Mass Parameters
- Sample Preparation: Prepare a 10 mg/mL solution of polymer in tetrahydrofuran (THF). Prepare a 20 mg/mL solution of DHB in THF with 0.1% TFA.
- Mixing/Spotting: Mix the polymer and matrix solutions at a 1:10 (v/v) ratio. Spot 1 µL of the mixture onto a stainless steel MALDI target and allow to dry.
- Acquisition: Acquire mass spectra in positive linear or reflection mode. Calibrate using a polymer standard of known mass (e.g., PEG).
- Data Analysis:
- Calculate Mn, Mw, and Đ from the isotopic distribution.
- Identify the mass difference between adjacent peaks to determine the monomer repeat unit(s) (e.g., 72 Da for lactide, 58 Da for glycolide).
- Identify the mass of the base peak to propose end-group combinations (e.g., H/OH, Na⁺ adduct).
Part B: NMR Analysis for Microstructure
- Sample Preparation: Dissolve ~15 mg of the same batch of polymer in 0.7 mL of CDCl3.
- Acquisition:
- Run a standard ¹H NMR spectrum (500 MHz). Integrate resonances to determine lactide:glycolide ratio (δ ~5.2 ppm for lactide methine, δ ~4.8 ppm for glycolide methylene).
- Run a ¹³C NMR spectrum. Analyze the carbonyl region (δ ~165-175 ppm). Splitting patterns indicate sequence distribution (e.g., lactide-lactide (LL), lactide-glycolide (LG) dyads).
- For tacticity, run a high-resolution ¹H NMR of the lactide methine region. The quartet splitting pattern indicates meso (m) or racemic (r) dyads.
- Data Integration:
- Use the absolute Mn from MALDI to convert NMR integration ratios into absolute numbers of end-groups and monomer units per chain.
- Correlate the proposed end-group masses from MALDI with the chemical shifts observed in the ¹H NMR spectrum (e.g., -OH, initiator fragments).
Protocol 3.2: Protocol for Biopolymer/Oligosaccharide Sequencing
Objective: Sequence a heparin-derived oligosaccharide.
Materials: Oligosaccharide sample, 2,4,6-trihydroxyacetophenone (THAP) matrix, cation exchange resin, D2O.
Part A: MALDI-TOF MS for Chain Length and Sulfation Degree
- Desalting: Pass sample through a mini-column of cation exchange resin (Na⁺ form). Elute with water.
- Sample Prep: Mix desalted sample with THAP matrix (10 mg/mL in 1:1 ethanol/water). Spot and dry.
- Acquisition: Acquire spectra in negative linear mode.
- Data Analysis: Determine chain length (dp) from m/z spacing (~Δm 300 Da for disaccharide). Calculate number of sulfo groups (Δm 80 Da per NaSO3) from mass deficit relative to fully sulfated species.
Part B: 2D NMR for Sugar Linkage and Sequence
- Sample Preparation: Lyophilize the desalted sample and dissolve in 99.9% D2O.
- Acquisition:
- Run ¹H-¹³C HSQC to assign anomeric proton/carbon signals.
- Run ¹H-¹H TOCSY to identify spin systems for individual sugar residues.
- Run ¹H-¹H NOESY or ¹H-¹³C HMBC to identify inter-residue linkages through through-space or long-range coupling correlations.
- Data Integration:
- Use the exact mass and sulfation count from MALDI to constrain the possible structures modeled from NMR data.
- The combined dataset allows building a specific sequence: [Sugar A-(linkage 1)-Sugar B-(linkage 2)-Sugar C] with known sulfation positions and chain length.
Visualizations
Title: Integrated MALDI and NMR Polymer Analysis Workflow
Title: Technique Selection Logic for Polymer Analysis
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Coupled MALDI-NMR Polymer Analysis
| Item Name | Function/Application | Critical Notes |
|---|---|---|
| DHB (2,5-Dihydroxybenzoic Acid) | Universal MALDI matrix for polar polymers (polyesters, polyethers). | Promotes soft ionization. Use with cation dopants (e.g., NaTFA, KTFA) for consistent adduct formation. |
| DCTB (trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile) | "Cool" matrix for polymers prone to fragmentation (e.g., PMMA, polystyrene). | Reduces in-source decay, yielding clearer spectra for mass analysis. |
| Trifluoroacetic Acid (TFA) 0.1% | Additive in matrix solution. | Protonates analytes and improves co-crystallization and homogeneity of the sample spot. |
| Deuterated Chloroform (CDCl3) | Primary NMR solvent for synthetic polymers. | Provides deuterium lock signal. Must be anhydrous for moisture-sensitive polymers (e.g., polyesters). |
| Deuterated Dimethyl Sulfoxide (DMSO-d6) | NMR solvent for polar, less-soluble polymers (e.g., polyamides, polysaccharides). | High boiling point, allows for elevated temperature NMR to increase polymer solubility. |
| Cation Exchange Resin (Na⁺ form) | Desalting/purification of samples (esp. biopolymers) pre-MALDI. | Removes adventitious salts that suppress ionization and cause peak broadening. |
| PEG or PMMA Calibration Standards | External calibration of MALDI-TOF MS. | Use a standard with chemistry and mass range similar to the analyte for best accuracy. |
| Tetramethylsilane (TMS) or solvent residual peak | Internal chemical shift reference for NMR. | TMS is added at δ 0.00 ppm. The solvent residual peak (e.g., CHCl3 at 7.26 ppm in ¹H) is commonly used. |
Within the context of a broader thesis on the MALDI-TOF MS method for polymer molecular weight research, it is imperative to compare it to its primary alternative, Electrospray Ionization Mass Spectrometry (ESI-MS). This analysis is critical for researchers, scientists, and drug development professionals selecting the optimal tool for polymer characterization, which impacts material properties and drug delivery system performance.
Core Comparison: MALDI-TOF MS vs. ESI-MS
Table 1: Direct Comparison of Key Characteristics
| Feature | MALDI-TOF MS | ESI-MS |
|---|---|---|
| Ionization Principle | Soft ionization via laser ablation of analyte embedded in a UV-absorbing matrix. | Soft ionization via electrospray creating charged droplets in a strong electric field. |
| Typical Mass Range | High (up to 1,000,000 Da for synthetic polymers). Ideal for high-mass species. | Lower (typically up to 70,000 Da under standard conditions). Better for lower mass polymers. |
| Mass Accuracy | Moderate to High (external calibration). ~100-500 ppm. | Very High (internal calibration possible). <10 ppm with high-resolution instruments. |
| Polydispersity (Đ) Analysis | Provides direct, visual MWD from spectrum. Can underestimate Đ for broad distributions due to detector saturation and discrimination. | Provides multiple charge states; requires deconvolution. Can better represent broad distributions. |
| Sample Complexity | Low tolerance for salts, additives. Requires clean samples and matrix selection. | Higher tolerance for buffers and additives. Direct infusion of solutions possible. |
| Throughput | High (sample plates hold 10s-100s of spots). | Lower (typically sequential sample introduction). |
| Quantitative Ability | Semi-quantitative; suffers from ionization bias and detector saturation. | More quantitative; linear response over a wider concentration range. |
| Polymer End-Group Analysis | Excellent due to single-charge-state peaks and high mass range. | Can be complicated by multiple charge state envelopes but provides confirmation. |
| Commonly Analyzed Polymers | PMMA, PS, PEG, Polyesters, PEGylated proteins. | Polyethers, PEG, Synthetic peptides, Protein-polymer conjugates, Polyolefins (with special solvents). |
Table 2: Quantitative Performance Data Summary
| Parameter | MALDI-TOF MS | ESI-MS (Quadrupole/TOF or FT-ICR) |
|---|---|---|
| Mass Resolution (Typical) | 10,000 - 20,000 (Reflectron mode) | 20,000 - 100,000+ (FT-ICR up to 10^6) |
| Mass Accuracy (ppm) | 100 - 500 (with external calibration) | 2 - 10 (with internal calibration) |
| Useful Mn Range (Da) | 1,000 - 350,000 | 500 - 70,000 |
| Sample Consumption | ~1 pmol per spot | ~50-100 fmol/µL (flow rate dependent) |
| Analysis Time per Sample | ~1-3 minutes (including averaging) | ~5-10 minutes (including MS/MS method) |
Ideal Use Cases
- MALDI-TOF MS is ideal for: Rapid determination of Mn, Mw, and Đ for narrow-distribution polymers (Đ < 1.2); precise end-group analysis; high-throughput screening of polymer libraries; analysis of high molecular weight synthetic polymers and large block copolymers.
- ESI-MS is ideal for: Detailed structural characterization using tandem MS (MS/MS) to sequence polymeric chains or confirm end-groups; analysis of polymers in native solution environments (e.g., with buffers); analyzing polymers with broad distributions more accurately; coupling with liquid chromatography (LC-ESI-MS) for separation of complex mixtures.
Detailed Experimental Protocols
Protocol 1: MALDI-TOF MS Analysis of Poly(methyl methacrylate) (PMMA) Standards Objective: Determine the average molecular weight and dispersity of a narrow PMMA sample.
- Materials: PMMA sample (~10 mg/mL in THF), DCTB matrix (trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile) (~20 mg/mL in THF), Sodium Trifluoroacetate (NaTFA) cationizing agent (10 mg/mL in THF), Stainless steel MALDI target plate.
- Sample Preparation (Dried-Droplet Method): a. Mix solutions in a vial with a volume ratio of Sample : Matrix : Cationizer = 5 : 10 : 1. b. Vortex for 30 seconds. c. Pipette 1 µL of the mixture onto a target plate spot. d. Allow to dry at ambient temperature to form homogeneous crystals.
- Instrumental Parameters (Generic):
- Ion Mode: Positive, Reflection.
- Laser Wavelength: 337 nm (Nitrogen laser).
- Acceleration Voltage: 20 kV.
- Extraction Delay Time: 200 ns.
- Number of Shots per Spectrum: 500 (accumulated from random spots on the sample spot).
- Data Analysis: a. Calibrate the spectrum using an external PMMA standard of known mass. b. Identify the series of peaks corresponding to [M + Na]+ ions. c. Calculate Mn, Mw, and Đ using the instrument's polymer analysis software, integrating the entire isotopic distribution for each n value.
Protocol 2: ESI-MS Analysis of Poly(ethylene glycol) (PEG) with Tandem MS Objective: Confirm the end-group structure of a mono-methyl ether PEG (mPEG) and obtain a mass spectrum.
- Materials: mPEG sample (~0.1 mg/mL in 50:50 MeOH:H2O with 0.1% Formic Acid), Syringe pump or LC system, ESI-compatible solvents (MeOH, H2O, CH3CN).
- Sample Introduction: Infuse the sample solution directly via a syringe pump at a flow rate of 5-10 µL/min.
- Instrumental Parameters (Q-TOF or FT-ICR):
- Ion Mode: Positive.
- Source Temperature: 120°C.
- Desolvation Gas Flow: 500 L/hr.
- Capillary Voltage: 3.0 kV.
- Cone Voltage: 30 V (low for minimal fragmentation).
- Mass Range: m/z 500 - 2000.
- For MS/MS: Select the [M+NH4]+ or [M+Na]+ ion of a specific oligomer. Use collision-induced dissociation (CID) with collision energy ramped from 15-35 eV.
- Data Analysis: a. Deconvolute the multiply charged spectrum ([M+nH]n+, [M+nNa]n+) using MaxEnt or similar software to obtain the zero-charge mass spectrum. b. For MS/MS, interpret the fragmentation pattern: loss of ethylene oxide units (44 Da) confirms the polymer backbone, while the mass of the terminal fragment identifies the end-group.
Visualization: Workflow and Logical Decision Path
Decision Workflow for Polymer MS Technique Selection
MALDI-TOF MS Polymer Analysis Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Polymer Mass Spectrometry
| Item | Function/Benefit | Common Examples (Polymer Analysis) |
|---|---|---|
| MALDI Matrices | Absorbs laser energy, facilitates analyte desorption and ionization with minimal fragmentation. | DCTB: Universal for synthetics (PMMA, PS). DHB: For polar polymers (PEG, polyesters). HABA: For higher mass polymers. |
| Cationization Agents | Promotes formation of [M+Cation]+ ions, simplifying spectra (MALDI) or controlling adduct formation (ESI). | NaTFA, KTFA: For synthetic polymers (Na+, K+ adducts). AgTFA: For polyolefins and non-polar polymers (Ag+ adduct). Ammonium Acetate: For ESI to promote [M+NH4]+. |
| ESI-Compatible Solvents | Volatile, promote droplet formation and desolvation in the ESI source. | LC-MS Grade MeOH, ACN, H2O. Mixtures with 0.1% FA or AA to assist protonation. Dichlorobenzene/MeOH for challenging polymers like polyolefins. |
| Polymer Standards | Critical for external calibration (MALDI) and validating method performance. | Narrow dispersity PMMA, PS, PEG with certified Mn values. |
| Liquid Chromatography Systems | Hyphenated with ESI-MS to separate polymer mixtures by end-group, composition, or size (LC-CC) prior to MS analysis. | Reversed-Phase C18 Columns (for low-mass oligomers). Size-Exclusion Columns (coupled to ESI-MS via split-flow). |
Within a broader thesis on MALDI-TOF MS for polymer molecular weight research, validation is paramount. This document provides application notes and detailed protocols for three pillars of validation: using well-defined polymer standards, conducting intra-laboratory reproducibility studies, and performing inter-laboratory comparisons. These practices are critical for generating reliable, comparable, and publication-quality data on molecular weight (Mn, Mw, PDI) and polymer structure.
Table 1: Common Polymer Standards for MALDI-TOF MS Calibration
| Polymer Class | Specific Standard (Supplier Examples) | Typical m/z Range (Da) | Key Uses |
|---|---|---|---|
| Polystyrene (PS) | PS 2,000; 5,000; 10,000 (PSS, Fluka) | 1,000 - 20,000 | Broad calibration for synthetic hydrophobic polymers. |
| Poly(ethylene glycol) (PEG) | PEG 1,000; 5,000; 10,000 (Sigma) | 500 - 15,000 | Calibration for polar polymers; assessment of cation adduction. |
| Poly(methyl methacrylate) (PMMA) | PMMA 3,000; 7,000 (PSS) | 1,500 - 15,000 | Calibration for polyacrylates; evaluation of fragmentation. |
| Proteins (for TOF) | Insulin, Cytochrome C, Myoglobin (Bruker) | 5,000 - 20,000 | High-mass accuracy verification; instrument tuning. |
Table 2: Key Metrics from a Hypothetical Inter-laboratory Comparison Study
| Laboratory | PS 5k Mn (Da) | PS 5k Mw (Da) | PDI | Reported Uncertainty (%, 1σ) |
|---|---|---|---|---|
| Lab A | 5,150 | 5,320 | 1.033 | ± 2.1% |
| Lab B | 5,310 | 5,510 | 1.038 | ± 3.5% |
| Lab C | 4,980 | 5,150 | 1.034 | ± 2.8% |
| Consensus Mean | 5,147 | 5,327 | 1.035 | |
| Standard Deviation | 165 | 180 | 0.003 |
Detailed Protocols
Protocol 1: Preparation and Use of Polymer Standards for Calibration
Objective: To generate a reliable external calibration curve for polymer molecular weight determination. Materials: See "Scientist's Toolkit" below. Procedure:
- Standard Selection: Select a minimum of three narrow dispersity (PDI < 1.1) standards from the same polymer class as the analyte, spanning the expected m/z range.
- Solution Preparation: Prepare individual stock solutions (~10 mg/mL) of each standard in a suitable solvent (e.g., THF for PS). Prepare a matrix solution (e.g., DCTB, 20 mg/mL in THF).
- Sample Spotting: For each standard, mix the polymer solution, matrix solution, and cationizing agent (e.g., NaTFA, 10 mg/mL) in a volume ratio of 1:10:1 (polymer:matrix:salt). Vortex.
- Deposition: Apply 0.5-1 µL of the mixture to the MALDI target plate. Allow to dry under ambient conditions to form a homogeneous crystalline layer.
- Data Acquisition: Acquire spectra for each calibration standard using a consistent, non-discriminatory method (e.g., linear/reflector mode as appropriate, high laser power to ensure good S/N, ≥500 shots per spectrum).
- Peak Assignment: In the instrument software, manually assign the monoisotopic mass or the centroid of the isotope cluster for 3-5 peaks across the spectrum of each standard.
- Curve Generation: Input the known m/z values of the assigned oligomers (calculated from the repeating unit mass and end-groups) into the software. Generate a linear or quadratic calibration curve. Accept only curves with a correlation coefficient (R²) > 0.995.
Protocol 2: Intra-Laboratory Reproducibility Study
Objective: To assess the precision of the sample preparation and measurement process within a single laboratory. Procedure:
- Sample Design: Use a single, well-characterized polymer sample (e.g., PS 10k) as a test specimen.
- Replicate Preparation: Independently prepare six (6) separate sample spots on the same target plate, following Protocol 1 steps 2-4 precisely. Randomize their positions on the target.
- Automated Acquisition: Use an automated acquisition method to collect spectra from each spot using identical instrumental parameters (laser power, shot count, detection mode).
- Data Processing: Process all spectra with identical parameters: baseline subtraction, smoothing (if any), and peak detection thresholds. Apply the same calibration curve.
- Statistical Analysis: For each replicate, record Mn, Mw, and PDI. Calculate the mean, standard deviation (SD), and relative standard deviation (RSD%) for each parameter. An RSD% of < 5% for Mn is typically considered acceptable for polymer analysis.
Protocol 3: Inter-Laboratory Comparison (Ring Trial)
Objective: To evaluate the accuracy and transferability of a MALDI-TOF MS method across different instruments and operators. Procedure:
- Central Coordination: A lead laboratory prepares and homogenizes a large batch of two unknown polymer samples (e.g., a polyester and a polystyrene). Aliquots are distributed to participating labs.
- Shared Protocol: All labs receive a detailed, standardized protocol covering sample preparation (specifying matrix, solvent, ratios, spotting method), instrument acquisition settings, and data processing rules.
- Local Analysis: Each lab performs the analysis in triplicate (following Protocol 2 principles) using their local MALDI-TOF MS instrument.
- Data Submission: Labs submit raw spectra and calculated results (Mn, Mw, PDI, and associated uncertainties) to the lead lab.
- Consensus Analysis: The lead lab performs statistical analysis (see Table 2) to determine the consensus mean, between-lab standard deviation, and any outliers. Results are shared with participants to identify sources of systematic error (e.g., calibration differences, spot homogeneity).
Diagrams
Title: MALDI Polymer Analysis with Validation Loop
Title: Inter-Lab Comparison (Ring Trial) Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for MALDI Polymer Analysis Validation
| Item | Function & Rationale |
|---|---|
| Narrow Dispersity Polymer Standards (PS, PEG, PMMA) | Provide known m/z points for external calibration. Low PDI ensures sharp peaks for accurate centroid determination. |
| trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB) | A superior "universal" matrix for polymers. Reduces fragmentation, promotes even co-crystallization, and yields low background. |
| Sodium Trifluoroacetate (NaTFA) | A commonly used cationizing agent. Provides a consistent source of Na+ ions to form [M+Na]+ adducts, simplifying the mass spectrum. |
| HPLC-Grade Tetrahydrofuran (THF) | A common solvent for dissolving polymers, matrices, and salts. High purity prevents interference from contaminants. |
| Stainless Steel or Ground Steel MALDI Target Plates | The sample substrate. A clean, polished surface is critical for homogeneous sample crystallization. |
| Precision Micropipettes (0.5-10 µL range) | For accurate and reproducible volumetric delivery of sample mixtures to the target plate. |
| Quality Control (QC) Polymer Sample | A stable, in-house polymer sample with previously characterized molecular weight, run regularly to monitor system performance over time. |
1. Introduction Within the broader thesis investigating MALDI-TOF MS for polymer molecular weight (MW) characterization, a critical examination of its inherent limitations is paramount. This document details the mass-dependent biases in detection efficiency and defines the practical "sweet spot" for reliable analysis. These factors directly impact the accuracy of reported molecular weight distributions (MWDs), particularly for polydisperse samples common in drug delivery system development.
2. Core Biases: Mass-Dependent Detection Ion detection in MALDI-TOF MS is non-uniform across mass ranges. Key phenomena include:
- Ionization Efficiency Bias: Lower mass oligomers often ionize more readily than higher mass chains, especially when using common matrices like Dithranol or CHCA. This can lead to under-representation of high-mass species.
- Detection System Bias: Microchannel plate (MCP) detectors exhibit a velocity-dependent response. Larger, slower-moving ions produce weaker signals, further suppressing high-mass detection.
- Desorption/Desolvation Bias: Incomplete desolvation or fragmentation can occur differentially, favoring the detection of species within an optimal mass range.
Table 1: Quantitative Impact of Mass-Dependent Bias on Common Polymer Standards
| Polymer System | Nominal Mass Range (Da) | Apparent Mn (Da) via MALDI (Biased) | Actual Mn (Da) via GPC (Reference) | Observed Deviation |
|---|---|---|---|---|
| Polystyrene (PS) Narrow | 2,000 - 10,000 | 4,850 | 5,200 | -6.7% |
| Polyethylene glycol (PEG) Broad | 5,000 - 25,000 | 12,100 | 15,300 | -20.9% |
| Polymethyl methacrylate (PMMA) | 10,000 - 50,000 | 24,500 | 32,800 | -25.3% |
Data synthesized from current literature. Deviations are illustrative of typical trends.
3. Defining the Analytical "Sweet Spot" The "sweet spot" refers to the mass range where detection efficiency is optimal and most linear, typically between ~500 Da and ~20,000 Da for most commercial MALDI-TOF instruments with standard settings. Outside this range, significant quantitative errors in MWD metrics (Mn, Mw, Đ) occur.
Table 2: Characteristics of the MALDI-TOF MS "Sweet Spot"
| Parameter | Within "Sweet Spot" (e.g., 5k-15k Da) | Outside "Sweet Spot" (e.g., >30k Da) |
|---|---|---|
| Signal-to-Noise Ratio | High (>100:1) | Low to Moderate (<50:1) |
| Mass Accuracy | Excellent (< 50 ppm) | Degraded (> 200 ppm) |
| Detector Response | Near-Linear | Highly Suppressed |
| MWD Fidelity (Đ) | High Correlation with GPC | Poor Correlation, Narrowing Artifact |
4. Detailed Protocol: Assessing Detection Bias and Sweet Spot Range Objective: To empirically determine the mass-dependent detection bias and effective "sweet spot" for a specific MALDI-TOF MS instrument and polymer-matrix system.
Materials & Reagents:
- Narrow Dispersity Polymer Standards: A series covering a broad mass range (e.g., PEG or PS: 1k, 5k, 10k, 20k, 40k Da).
- Matrix: Suitable for polymer (e.g., Dithranol, Trans-2-[3-(4-tert-Butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB)).
- Cationization Agent: Trifluoroacetic acid salt (e.g., NaTFA, KTFA).
- Solvent: HPLC-grade Tetrahydrofuran (THF) or Chloroform.
Procedure:
- Sample Preparation: Prepare individual standard solutions (10 mg/mL in THF). Prepare matrix solution (20 mg/mL in THF). Prepare cationization agent solution (10 mg/mL in THF).
- Spotting: Use the dried-droplet method. For each standard, mix 10 µL polymer, 10 µL matrix, and 1 µL cationizer. Vortex and spot 1 µL onto the target plate. Allow to crystallize at room temperature.
- Instrument Calibration: Calibrate the instrument using a standard calibrant mix that brackets the expected mass range.
- Data Acquisition: Acquire spectra for each standard spot using identical instrumental settings (laser power, detector voltage, extraction delay). Use reflector mode for masses <10k Da, linear mode for >10k Da. Collect spectra from at least 10 different positions per spot.
- Data Analysis: For each standard, measure the total integrated area of the isotopic envelope for the most abundant charge state. Normalize this area by the theoretical molar concentration of polymer in the spot.
- Bias Curve Plotting: Plot the Normalized Signal Intensity (Y-axis) against the Known Mass of each standard (X-axis, logarithmic scale). The plateau region of this curve defines the instrument's effective "sweet spot."
5. Mitigation Strategies and Advanced Protocols Protocol: Expanding the Sweet Spot via High-Mass Detection Enhancement Objective: To improve the detection of high-mass polymers (>30 kDa). Key Modifications:
- Matrix Selection: Use a high-mass-friendly matrix like DCTB.
- Sample Preparation: Employ the layer method or electrospray deposition for more homogeneous co-crystallization.
- Instrument Settings: Increase linear detector voltage (within manufacturer limits). Optimize extraction delay time (pulsed ion extraction) for higher masses. Use only linear detection mode.
- Data Processing: Apply a mass-dependent correction factor derived from the bias curve in Section 4.
6. The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function & Role in Mitigating Bias |
|---|---|
| DCTB Matrix | Superior matrix for hydrophobic polymers, promotes desorption of high-mass ions with less fragmentation. |
| Silver Trifluoroacetate (AgTFA) | Powerful cationizing agent for polymers lacking heteroatoms, enhances ionization of polyolefins and polystyrenes. |
| Electrospray Deposition Device | Creates ultra-thin, homogeneous sample-matrix films, improving reproducibility and reducing mass discrimination. |
| Narrow Đ Calibration Kits | Pre-characterized polymer standards essential for constructing the detector bias curve and validating the "sweet spot". |
| GPC/SEC System with MALS | Provides orthogonal, bias-free molecular weight data for correlation and correction of MALDI-TOF results. |
Conclusion
MALDI-TOF MS stands as an indispensable, high-resolution tool for absolute polymer molecular weight determination and detailed structural analysis, particularly excelling in low dispersity systems and end-group characterization. While its methodological success hinges on meticulous sample preparation and cationization, its power is fully realized when used complementarily with SEC for dispersity and NMR for stereochemistry. For researchers in drug development, mastering MALDI-TOF MS enables precise characterization of polymer therapeutics, controlled-release matrices, and complex biomaterials. Future directions point towards increased automation, integration with chromatography (LC-MALDI), and advanced data processing with machine learning to handle ever-more complex polymeric architectures, solidifying its critical role in advancing polymer science for biomedical applications.